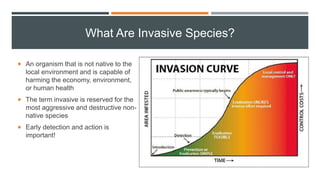
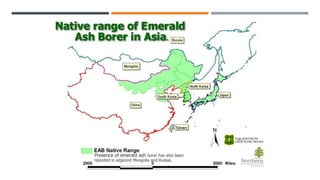
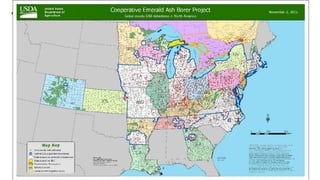
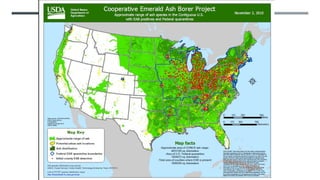
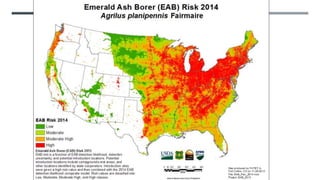
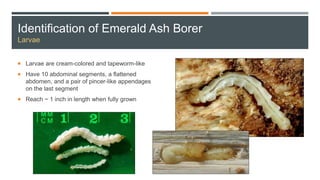
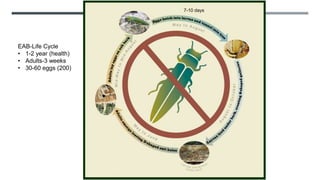
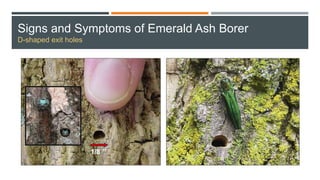
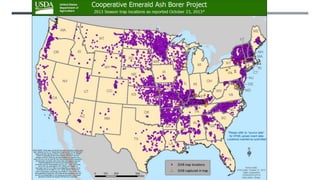
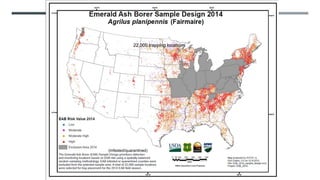
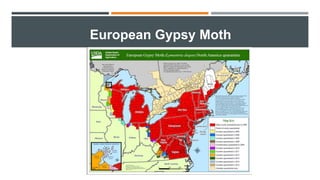
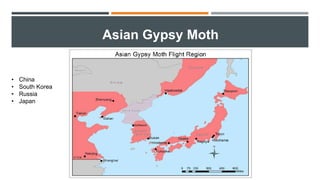
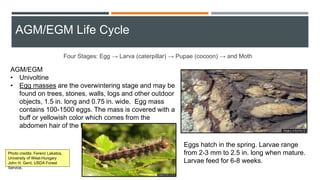
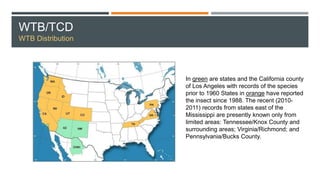
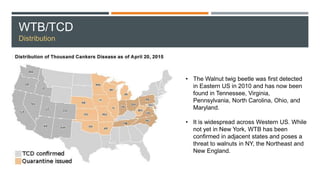
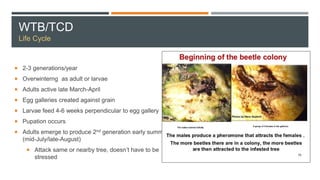
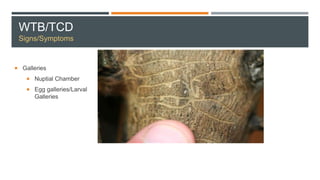
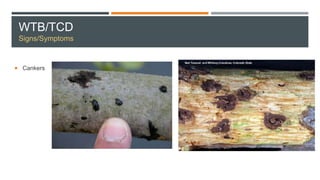
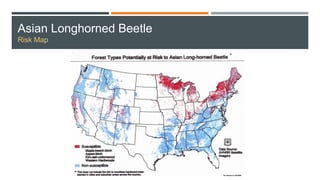
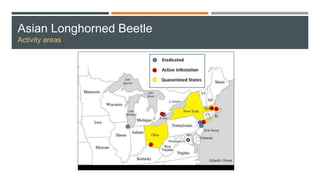
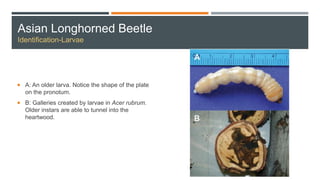
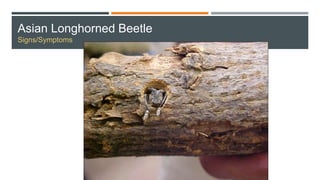
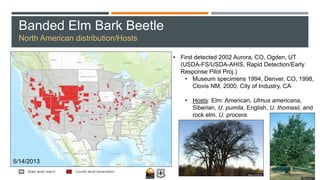
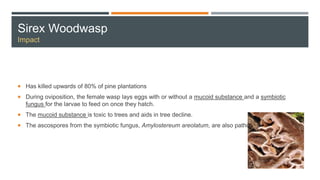
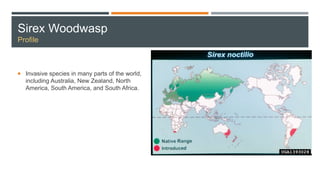
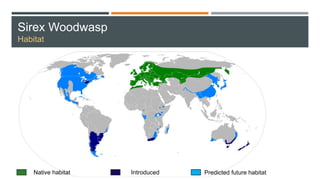
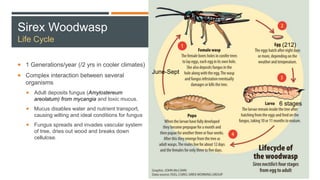
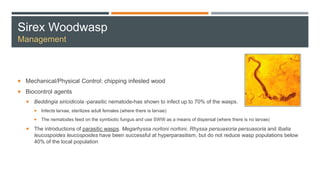
This document provides an overview of invasive insect species, focusing on their identification, impact, and management strategies, with a particular emphasis on the emerald ash borer, gypsy moths, walnut twig beetle, and Asian longhorned beetle. It outlines the threats these species pose to native trees, ecosystems, and economies, along with methods for monitoring and control. The importance of early detection and regulations on the movement of infested materials are highlighted to prevent further spread.