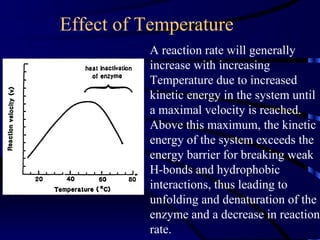
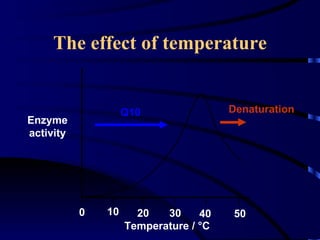
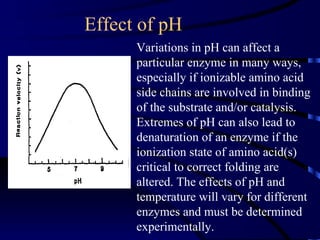
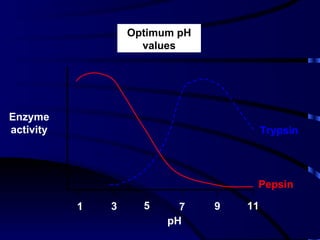
- Amylase, lipase, proteases added to laundry detergents
- Papain, bromelain added to meat tenderizers
- Lysozyme added to wound dressings
Diagnostic:
- Measuring enzyme levels in blood/urine to detect organ damage
- Measuring enzyme levels in blood to diagnose genetic disorders
Therapeutic:
- Enzyme replacement therapy for genetic disorders
- Enzymes as digestive aids or supplements
Research:
- Enzymes used as reagents in clinical assays and diagnostic kits
So in summary, enzymes play important roles in diagnostics, research, and therapeutics in medicine. Their catalytic properties are exploited for various applications.