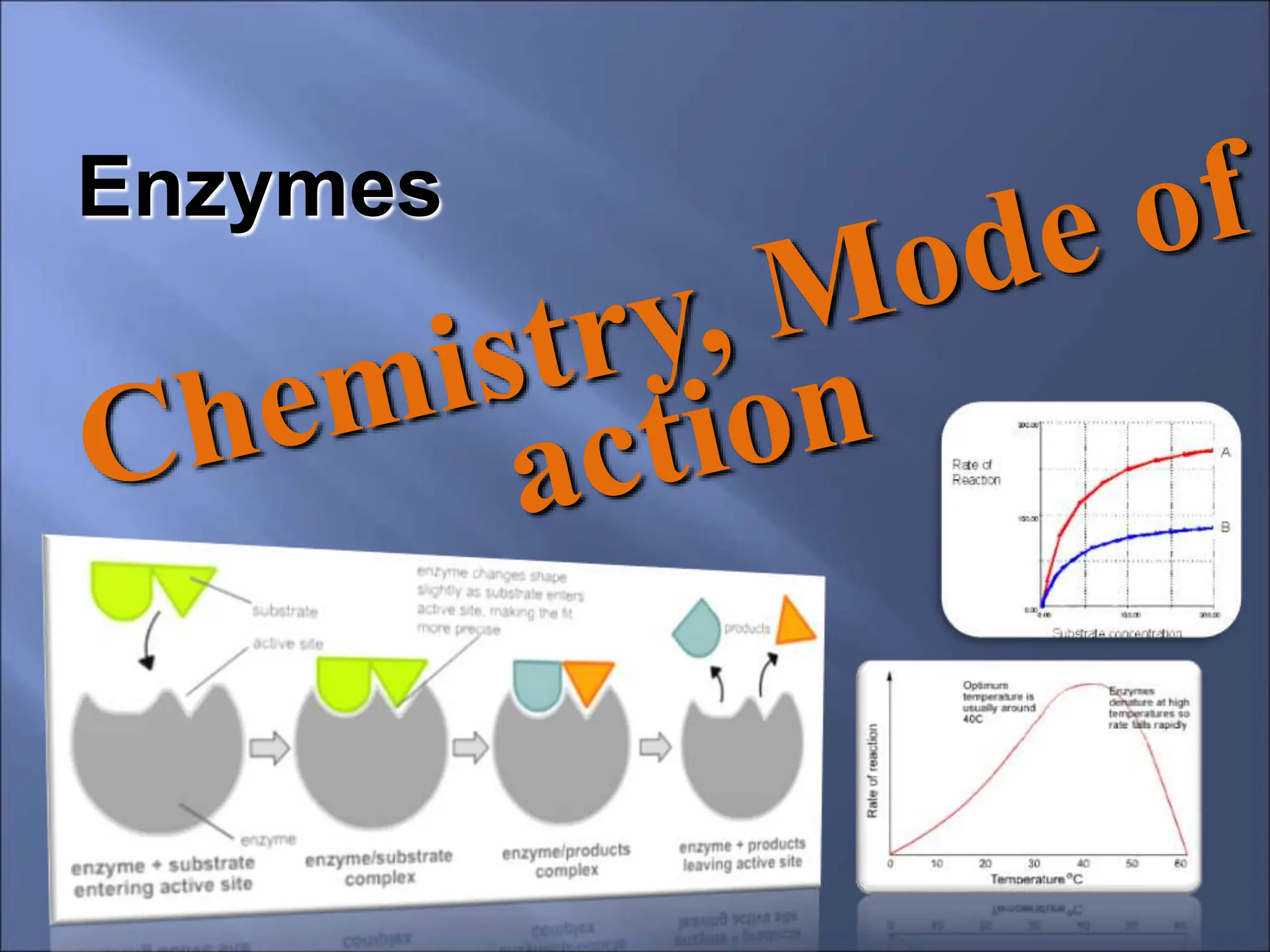
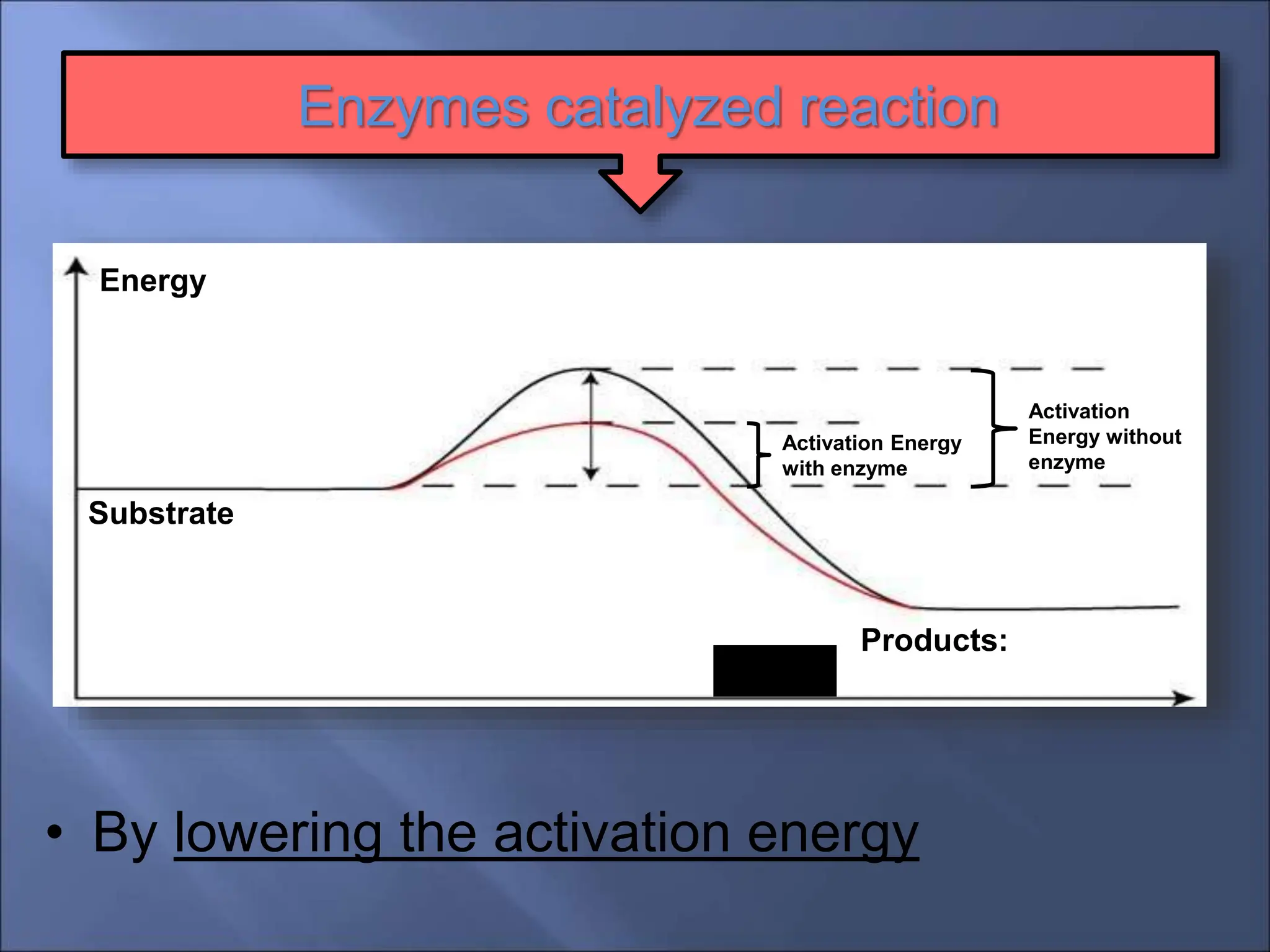
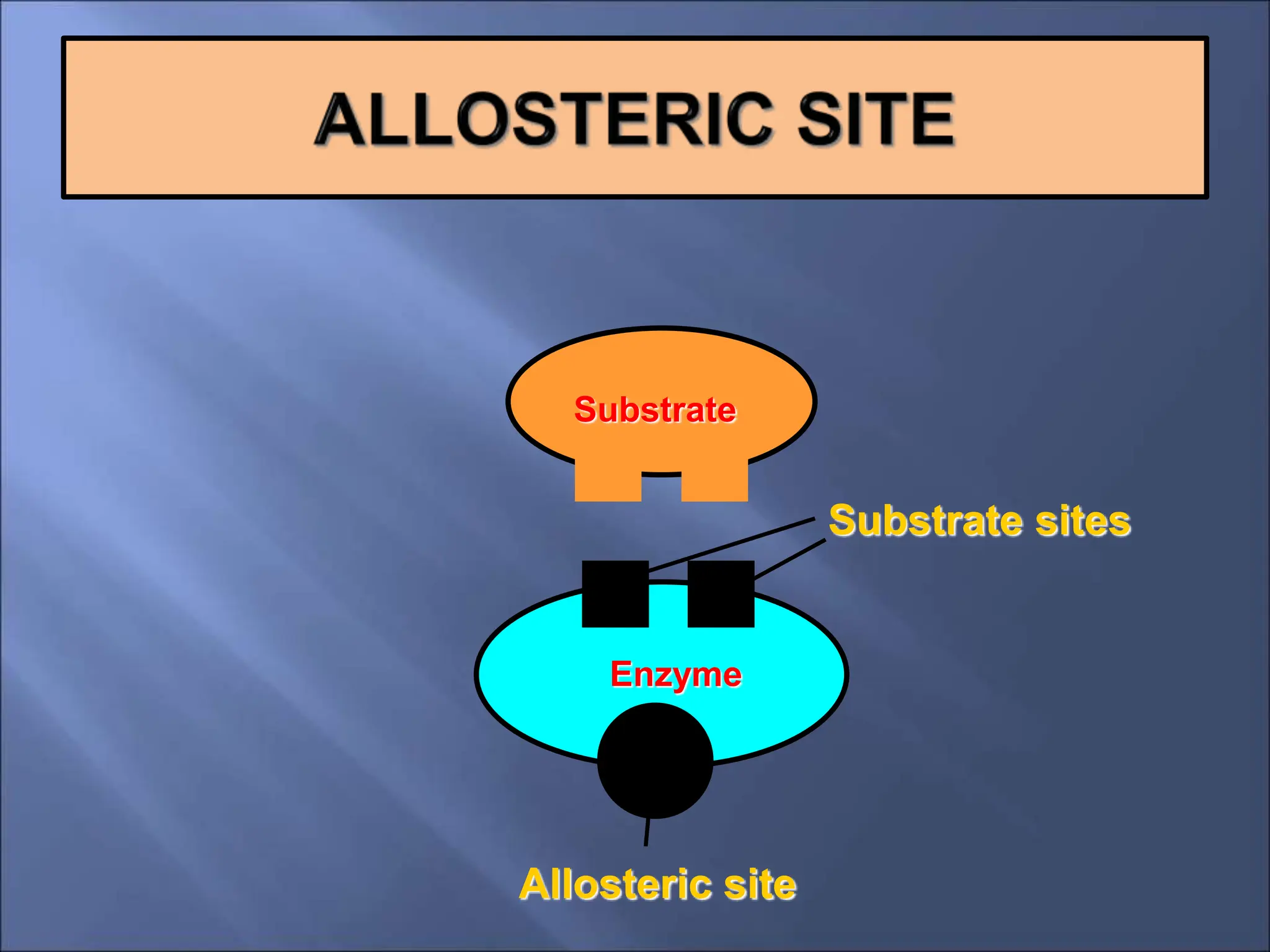
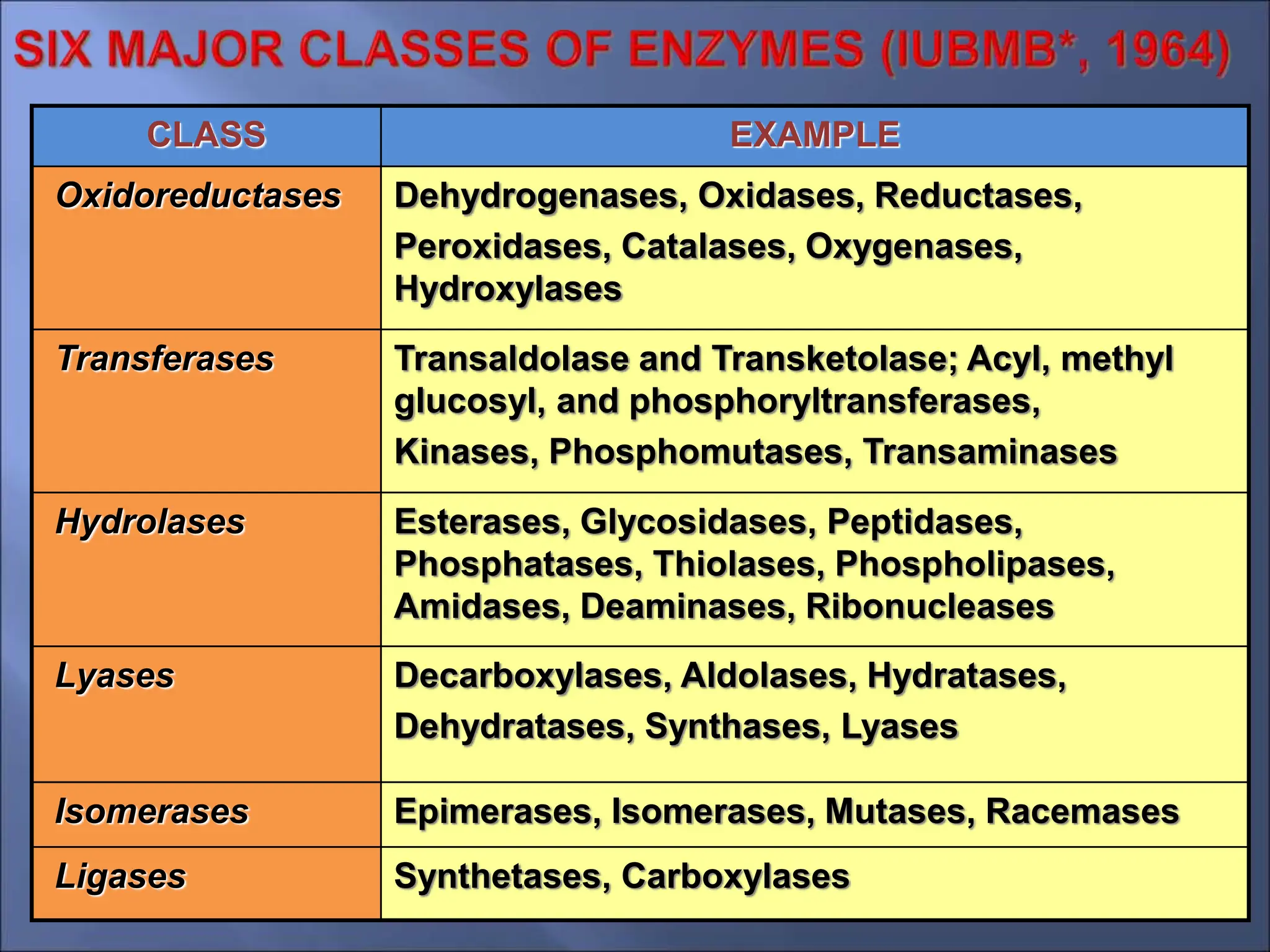
Enzymes are protein catalysts that accelerate chemical reactions by lowering their activation energy. They are highly specific and function most efficiently at their optimal temperature and pH. Some enzymes require cofactors like coenzymes to function. Enzyme activity can be regulated by inhibitors and allosteric regulation. Enzymes speed up reactions but are not consumed in the process, allowing them to catalyze many turns of a reaction.