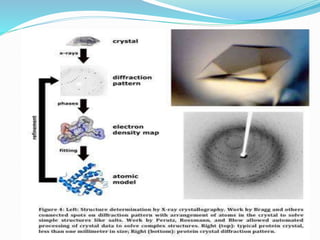
X-ray crystallography is a technique used to determine the atomic and molecular structure of crystals. X-rays are directed at a crystal and the diffraction pattern produced is analyzed to reveal the crystal structure. This information can be used to construct a 3D electron density map and determine atomic positions. Some important applications of X-ray crystallography include determining the structures of proteins, viruses like HIV, and using this information to develop new drugs.