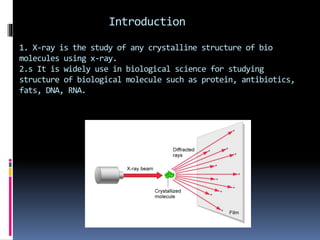
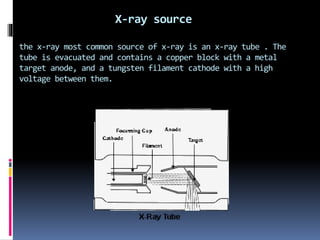
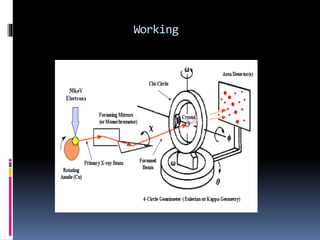
X-ray crystallography is a technique developed in the 17th century for studying the structure of biomolecules like proteins and DNA using high-energy X-ray beams. The process involves an X-ray diffractometer that includes components such as an X-ray source, monochromator, goniometer, and detector to capture diffraction patterns, which reveal the 3D arrangement of atoms in crystals. Applications of this technique extend to analyzing molecular structures, including those of various biomolecules and compounds like penicillin and insulin.