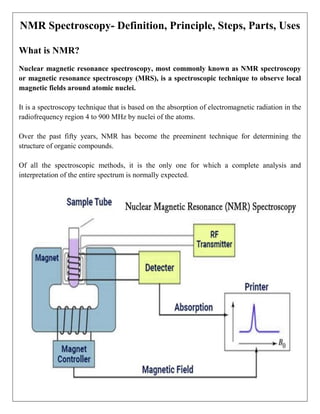
NMR spectroscopy, or nuclear magnetic resonance spectroscopy, is a technique used to observe local magnetic fields around atomic nuclei and has become vital for determining the structure of organic compounds. It operates on the principles of energy absorption and emission in the presence of a magnetic field, providing detailed information about molecular structure, dynamics, and environments. NMR finds applications in quality control, chemical structure analysis, and has advanced techniques for studying complex molecules and proteins.