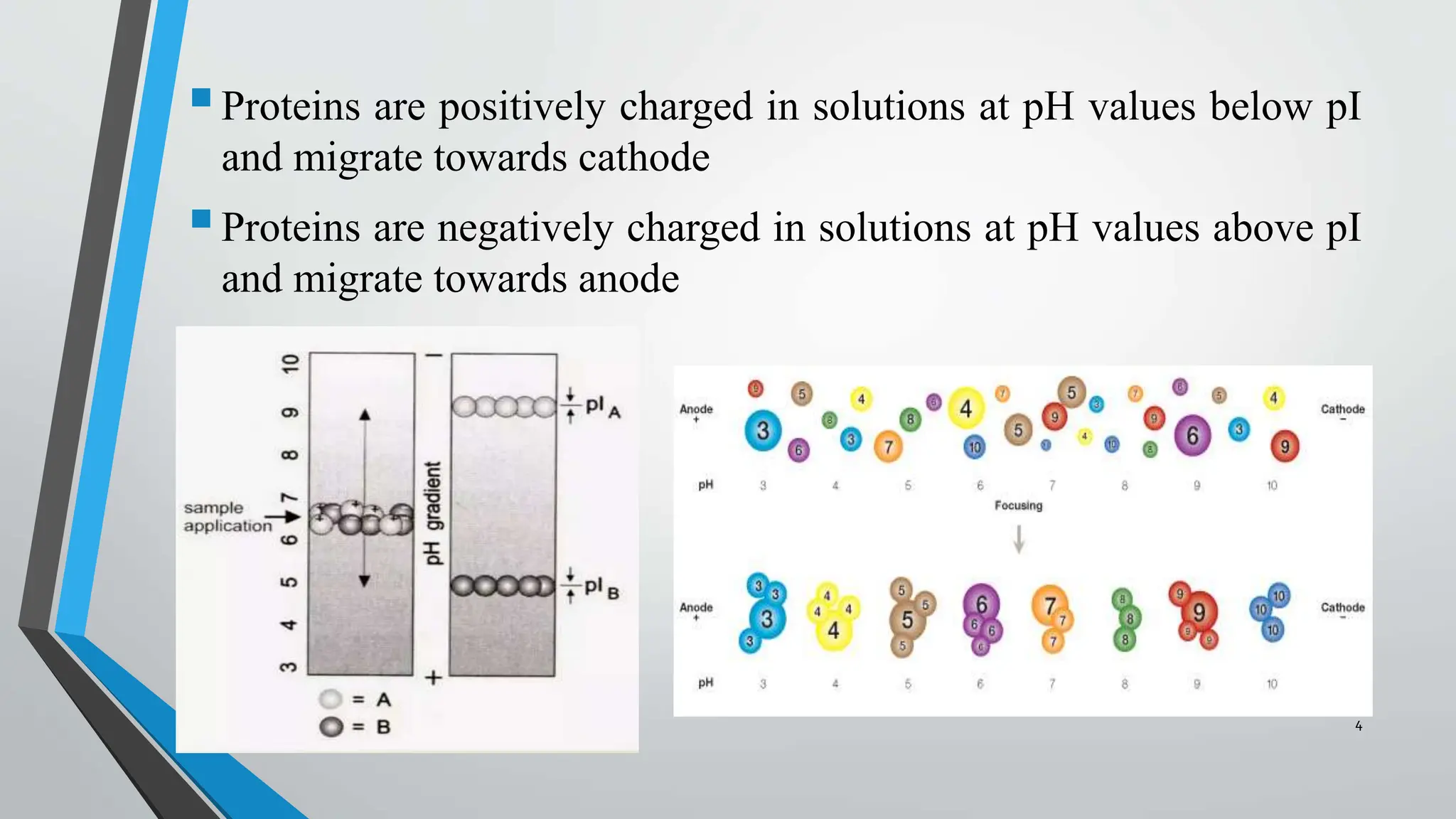
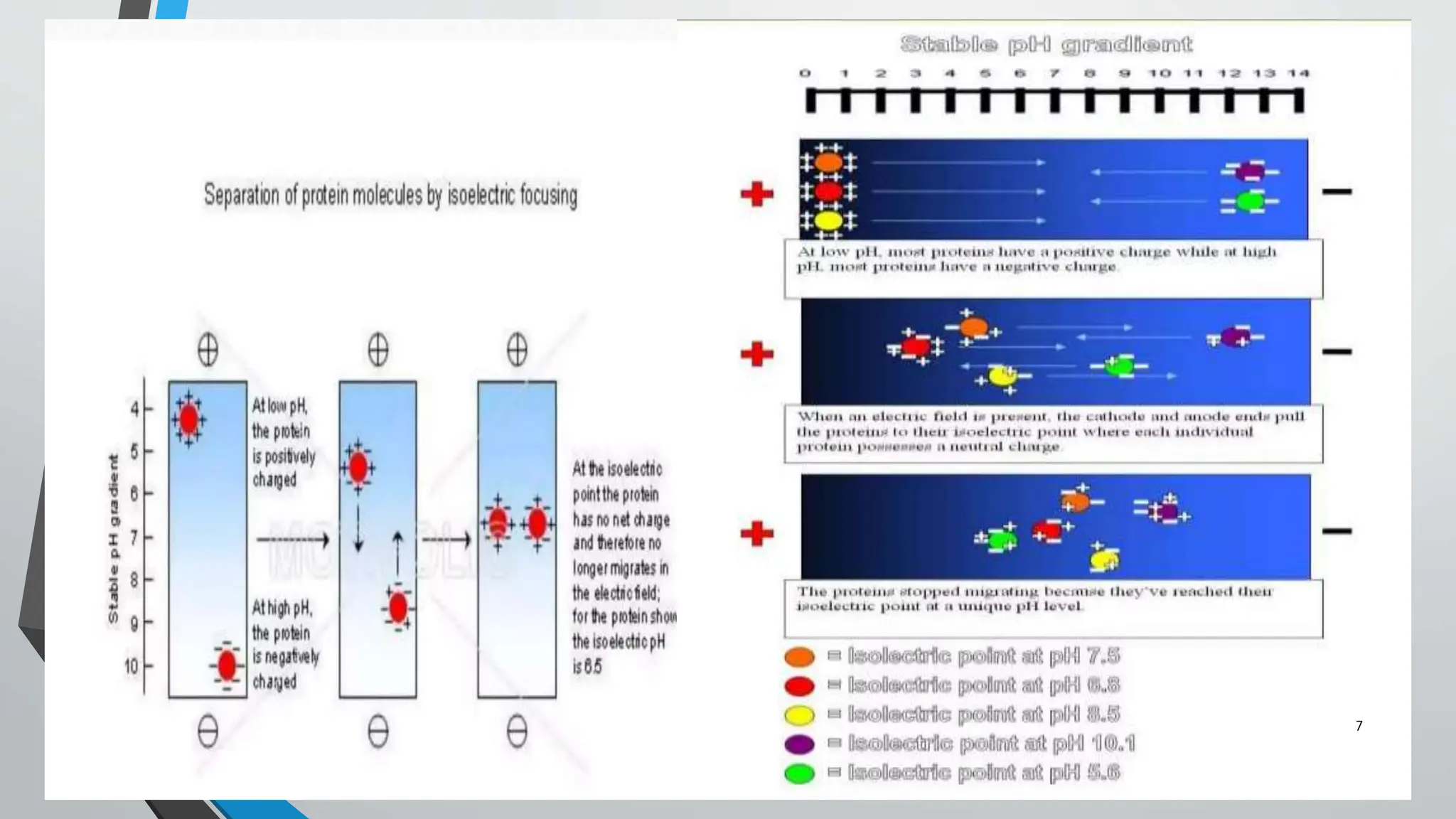
Isoelectric focusing (IEF) is an electrophoresis technique used to separate proteins based on their isoelectric point (pI), which is the pH at which the protein's net charge is zero. The process involves a pH gradient where proteins migrate until they reach their pI, resulting in well-defined bands that facilitate analysis and identification. IEF is widely utilized for quality control in therapeutic products, protein purification, and studying protein microheterogeneity.