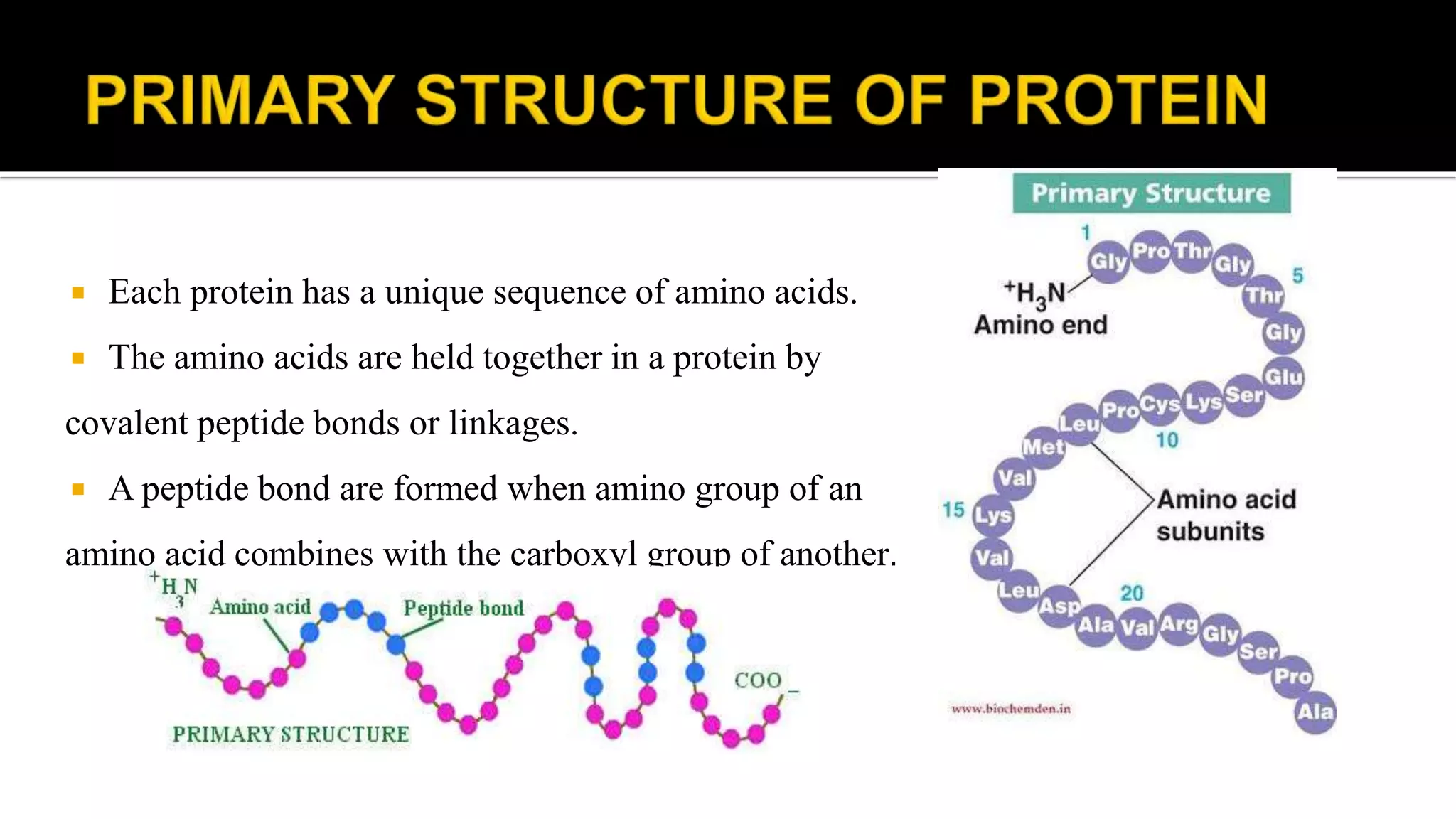
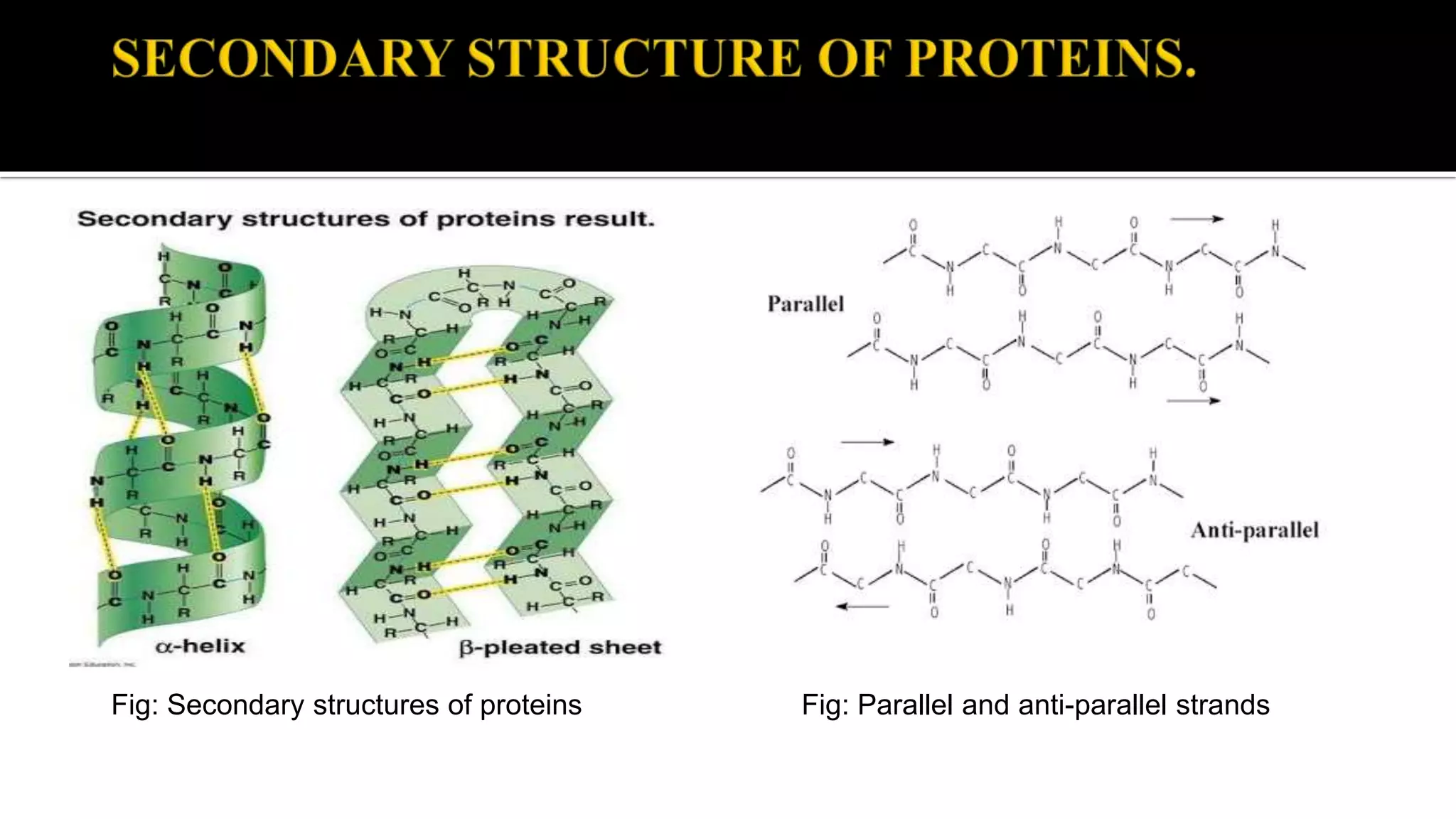
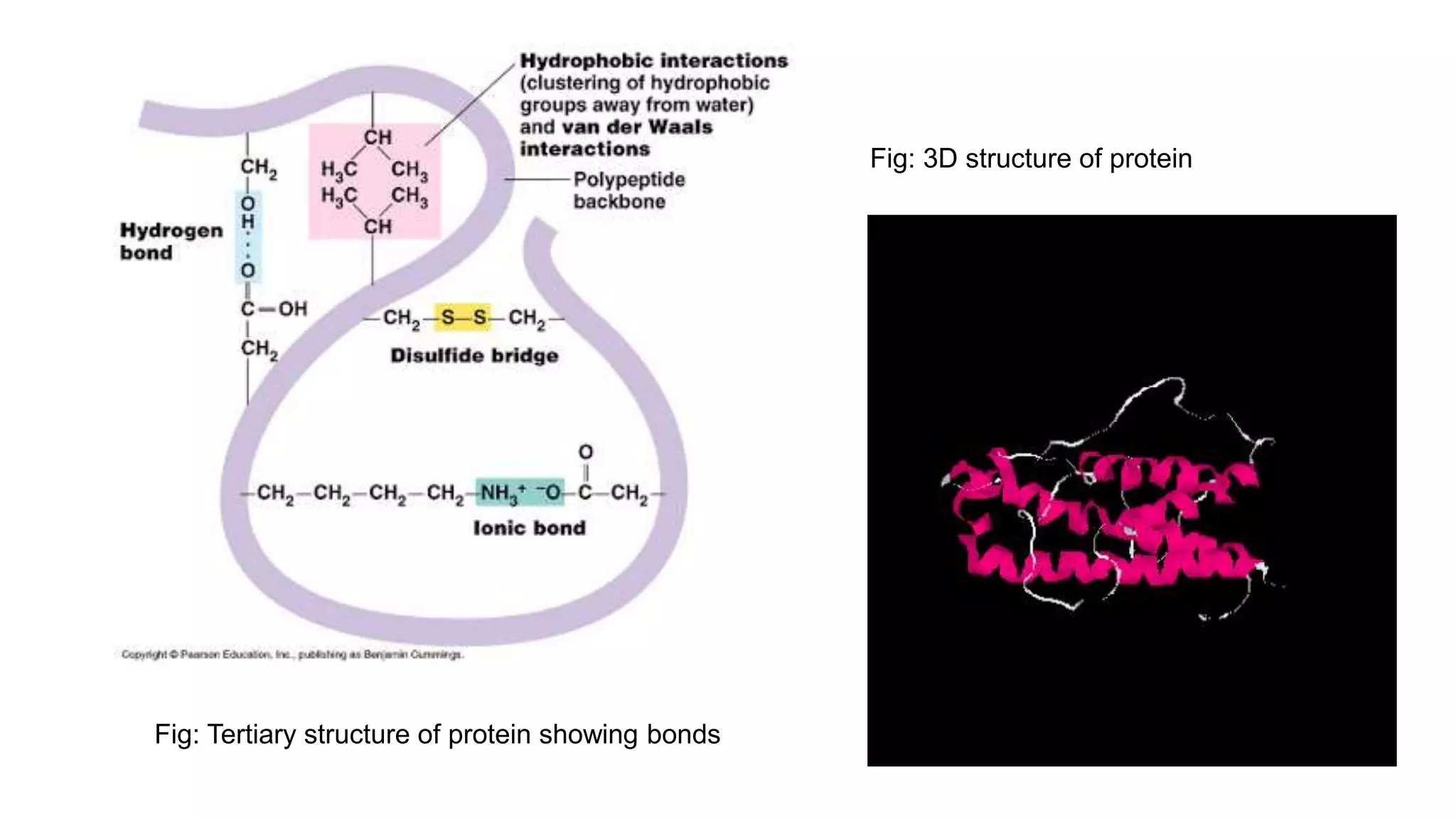
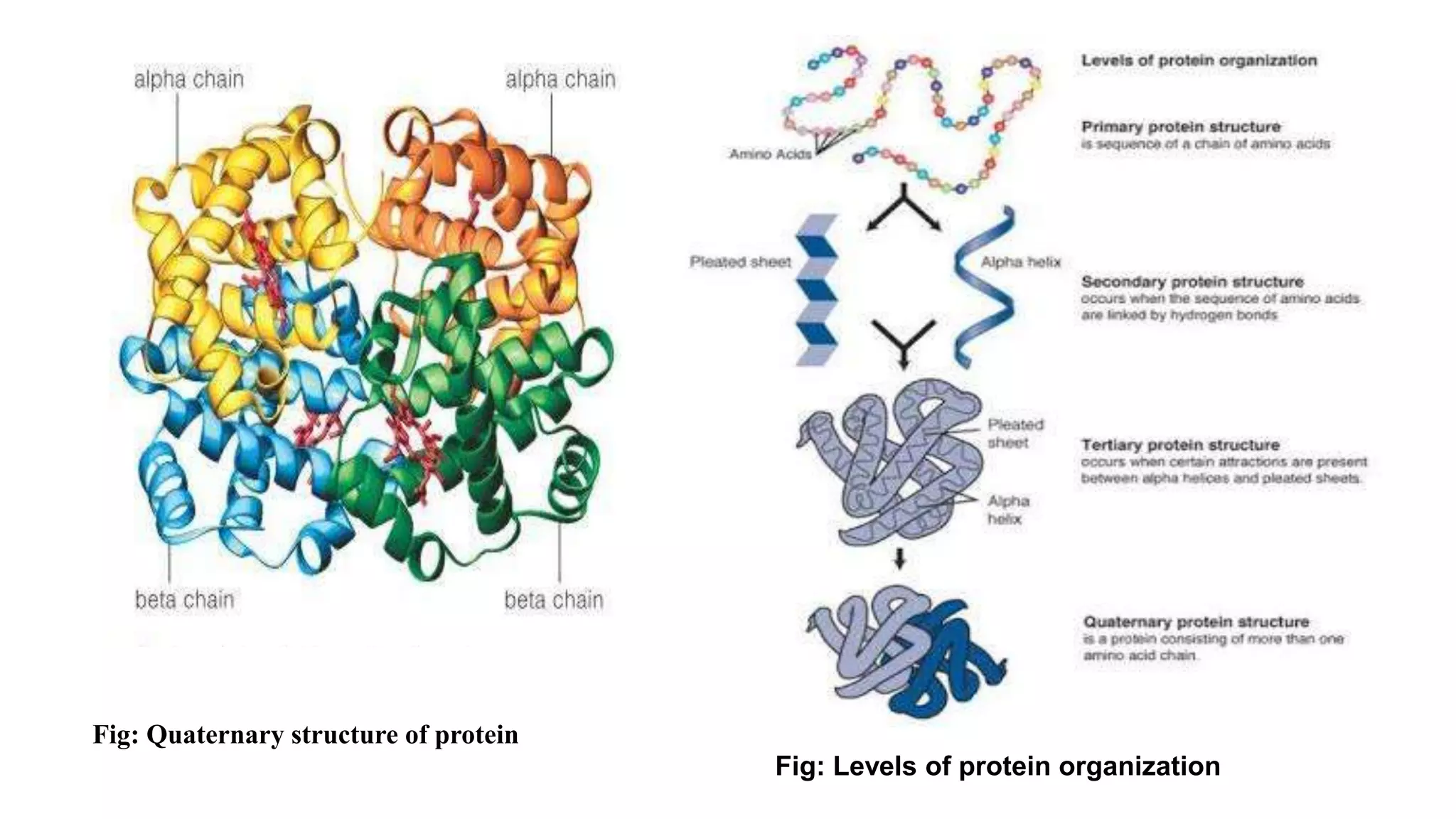
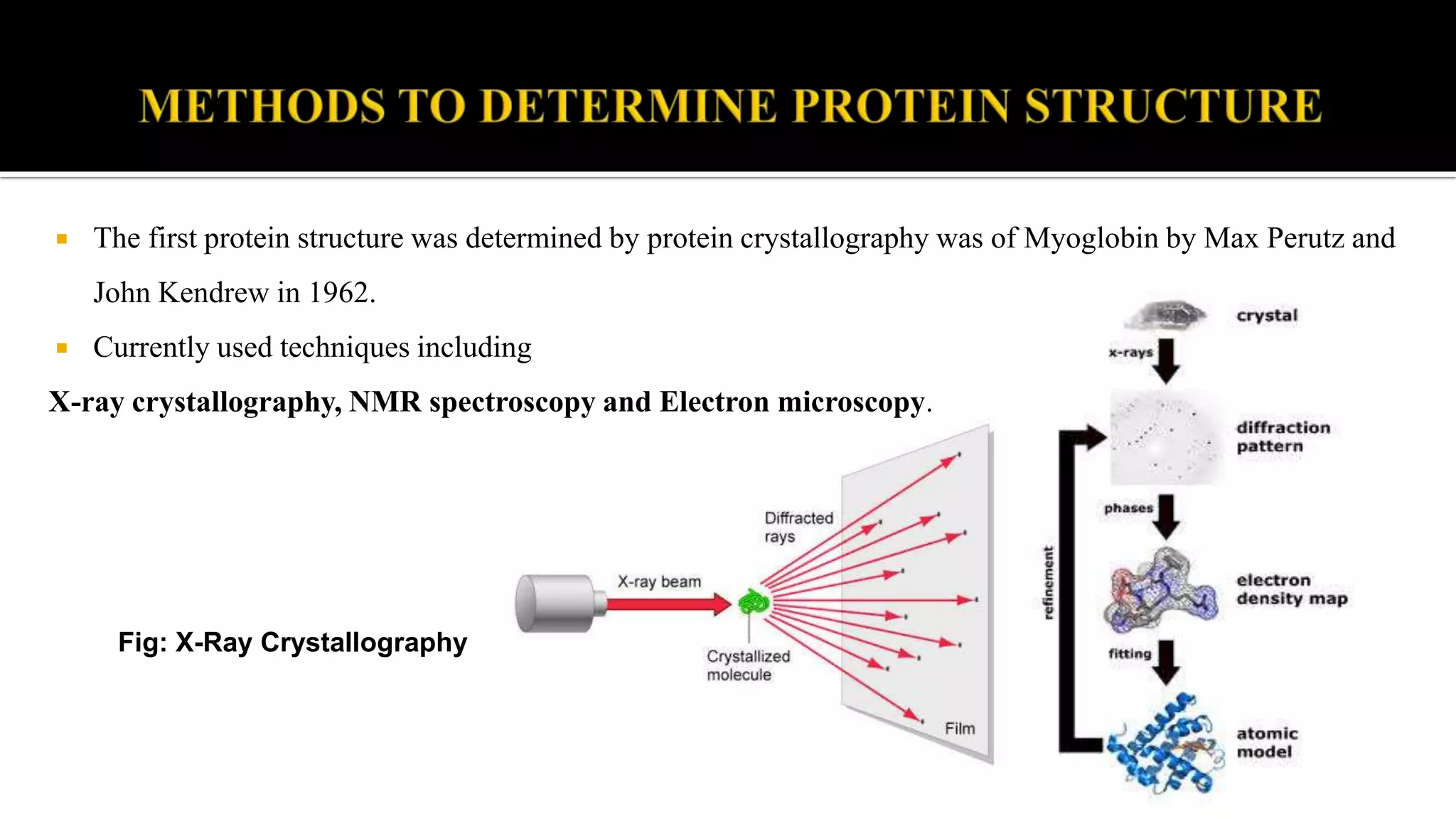
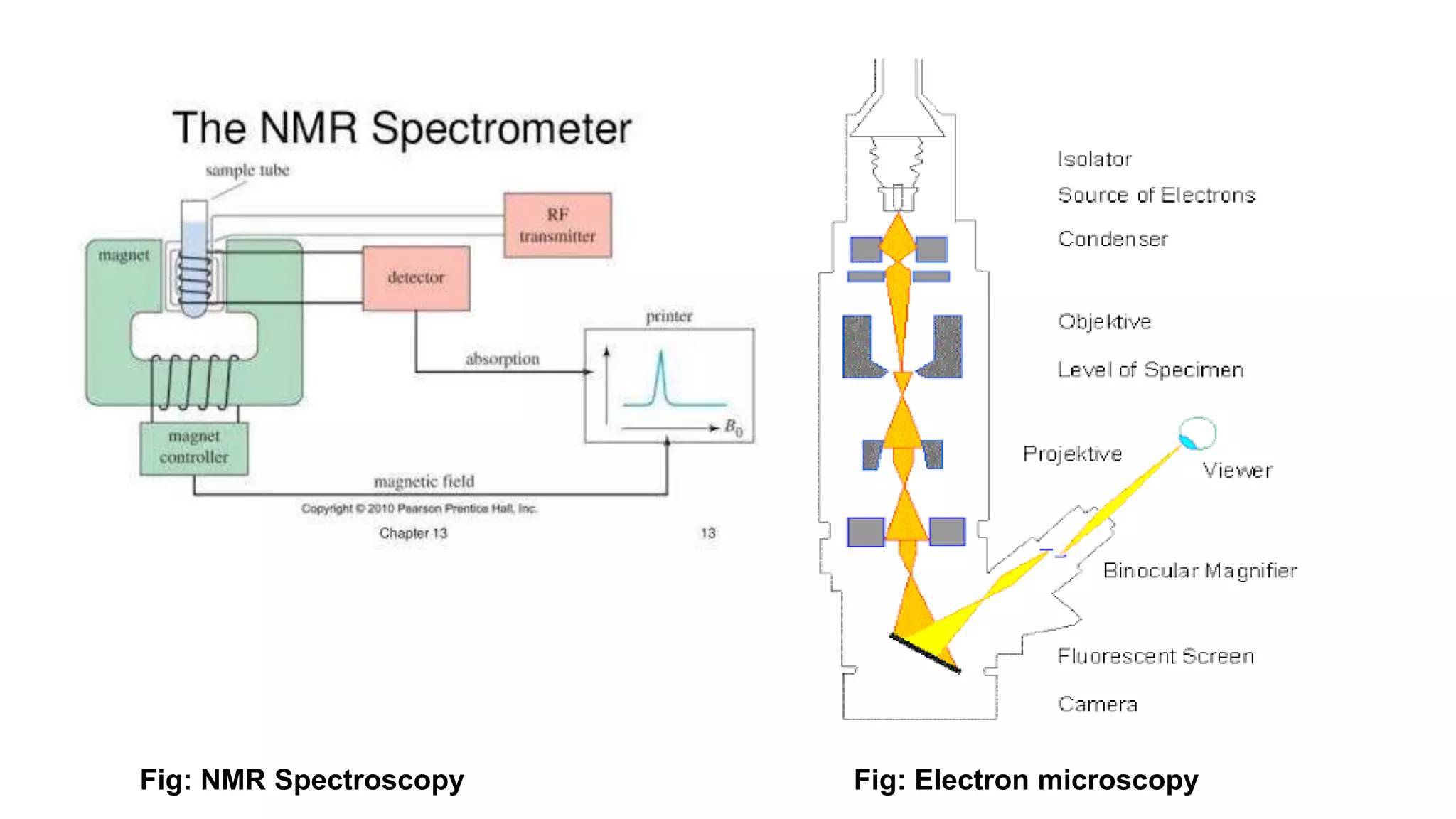
The document discusses the structural organization of proteins, detailing their primary, secondary, tertiary, and quaternary structures. It explains how proteins are made up of amino acids held together by peptide bonds and describes different techniques for determining protein structure, such as x-ray crystallography and NMR spectroscopy. Proteins are vital organic molecules in living organisms, serving numerous functions including enzymatic, hormonal, and structural roles.