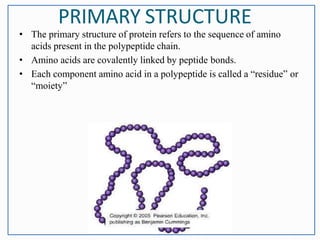
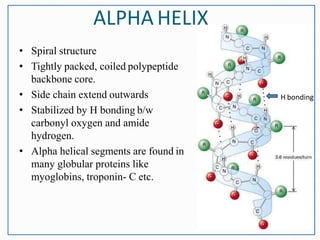
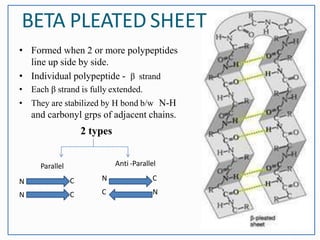
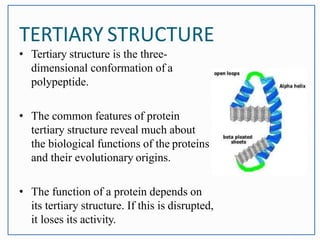
This document discusses the different levels of protein structure: primary, secondary, tertiary, and quaternary. The primary structure refers to the amino acid sequence. Secondary structure involves local folding into alpha helices or beta sheets. Tertiary structure describes the overall 3D shape of a single polypeptide chain. Quaternary structure refers to the arrangement of multiple polypeptide subunits in a multimeric protein. Recent discoveries are mentioned about determining the structures of specific proteins and learning more about their functions.