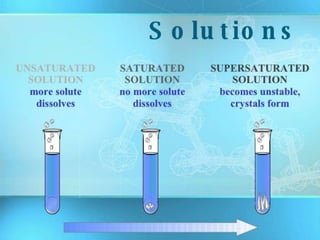
A solution is a homogeneous mixture of two or more substances where a solute is dissolved in a solvent. The concentration of a solution refers to the amount of solute dissolved in a fixed amount of solvent. Solutions can be dilute, containing a small amount of solute, or concentrated, containing a large amount of solute. Soft drinks are considered solutions but contain many unhealthy additives like sugar, artificial sweeteners, caffeine, carbon dioxide, preservatives, artificial flavors, colorings, and sodium. Long term consumption of soft drinks can increase risks of diseases, cancer, and other health issues.