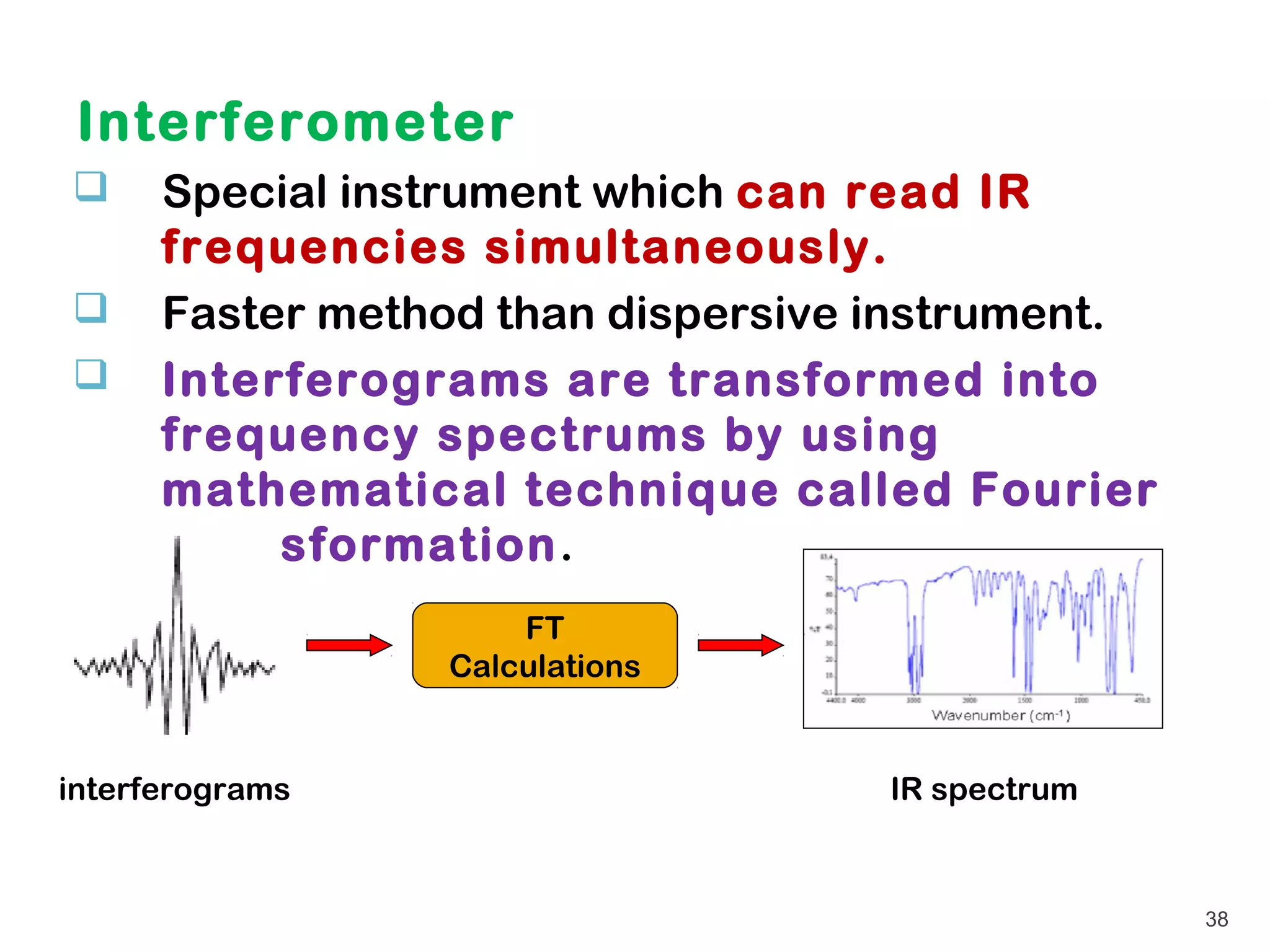
This document discusses infrared (IR) spectroscopy. It provides information on the basic principles of IR spectroscopy, sample preparation techniques, instrumentation including dispersive and Fourier transform IR spectrometers, data analysis and interpretation. Key points covered include how IR spectroscopy can be used to identify functional groups in molecules based on their characteristic absorption frequencies, and that each compound produces a unique IR spectrum that can be considered a "fingerprint" of its structure.