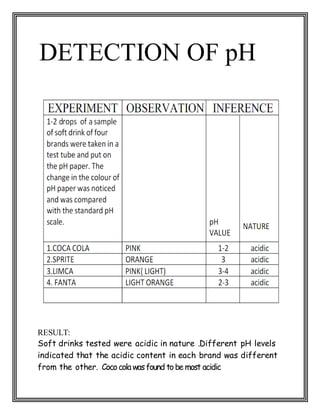
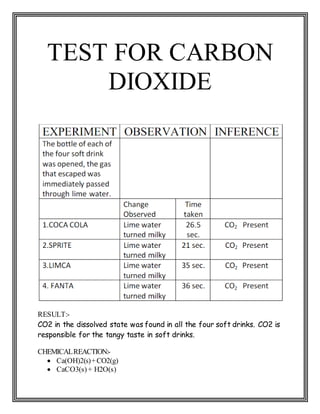
This chemistry project examines the contents and adverse effects of soft drinks. Through a series of tests, the student found that four popular soft drinks brands (Coca Cola, Sprite, Limca, Fanta) contain glucose, alcohol, sucrose, phosphate, and carbon dioxide. Coca Cola had the lowest pH (most acidic). While sugars provide energy, high intake through soft drinks can lead to health issues like diabetes and weight gain. Phosphoric acid damages tooth enamel and the acidic environment harms digestive organs. However, trace alcohol levels do not affect health. The project concludes that while soft drinks taste good, their acidic contents and high sugars have negative health consequences.