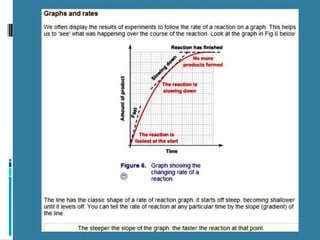
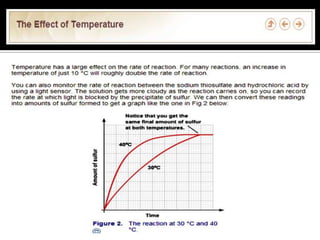
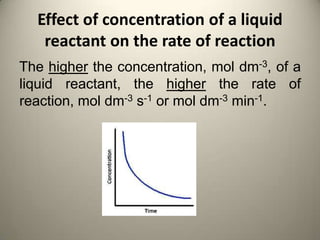
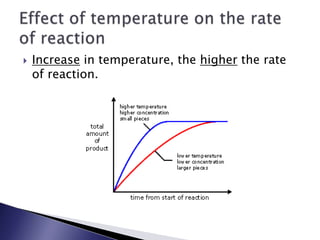
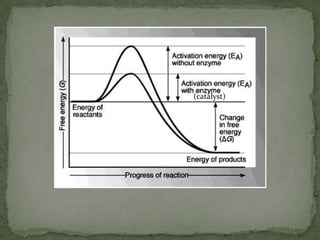
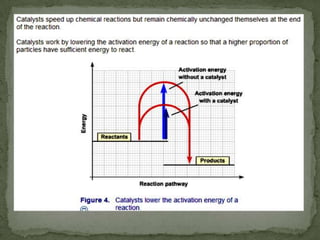
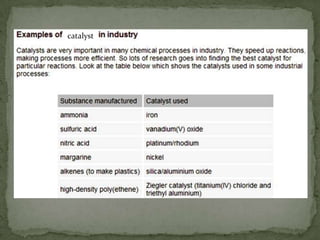
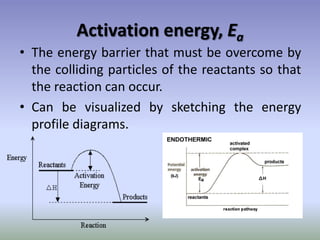
Rate of reaction is defined as the change in quantity of reactants or products per unit time. The average rate is calculated over an interval of time, while the instantaneous rate is the actual rate at a given time. Several factors affect the rate of reaction, including the total surface area and concentration of reactants, temperature, use of catalysts, and pressure for gaseous reactants. According to collision theory, the rate of reaction depends on the frequency and effectiveness of collisions between reactant particles, which must achieve the minimum activation energy and correct orientation.