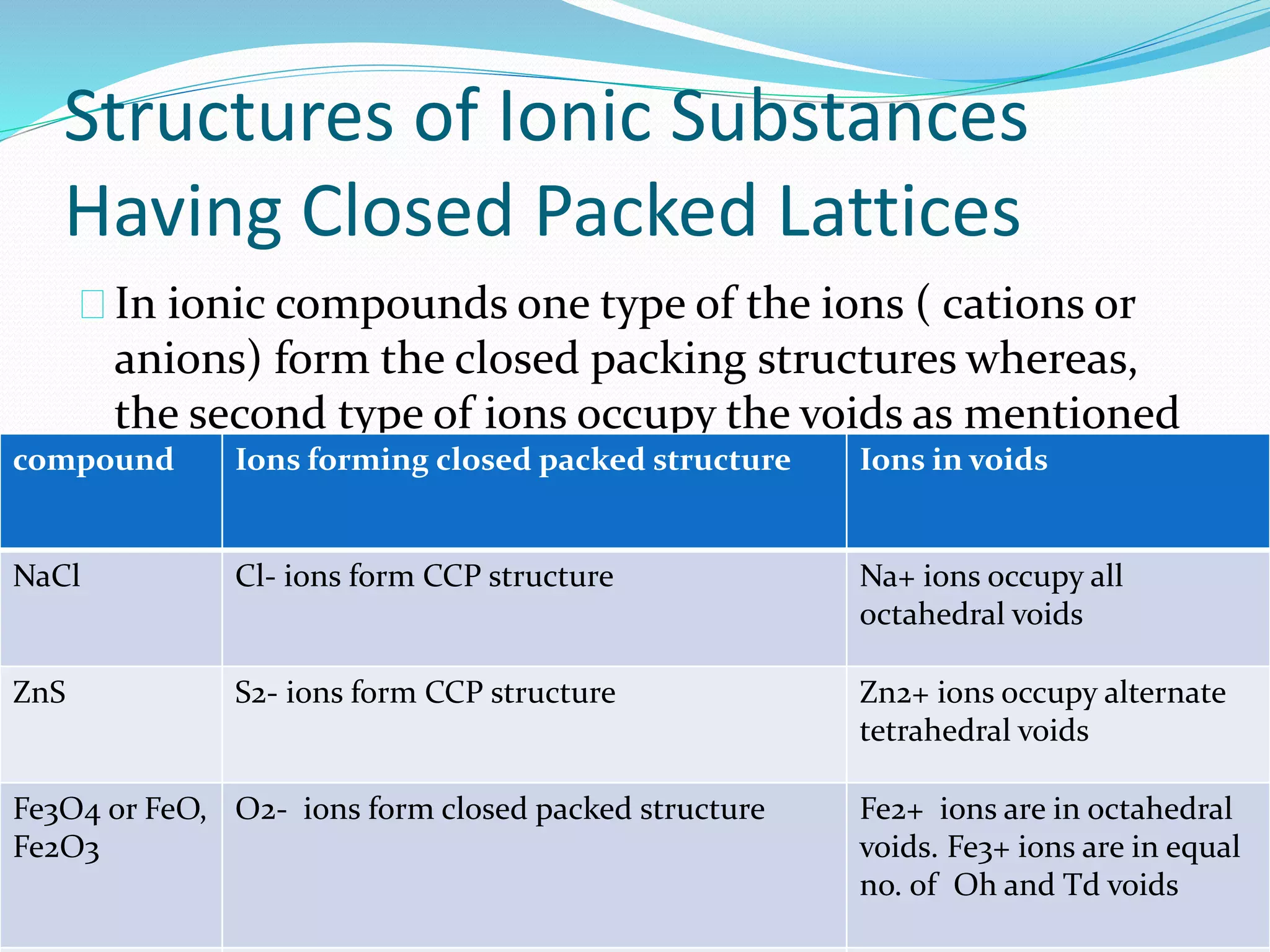
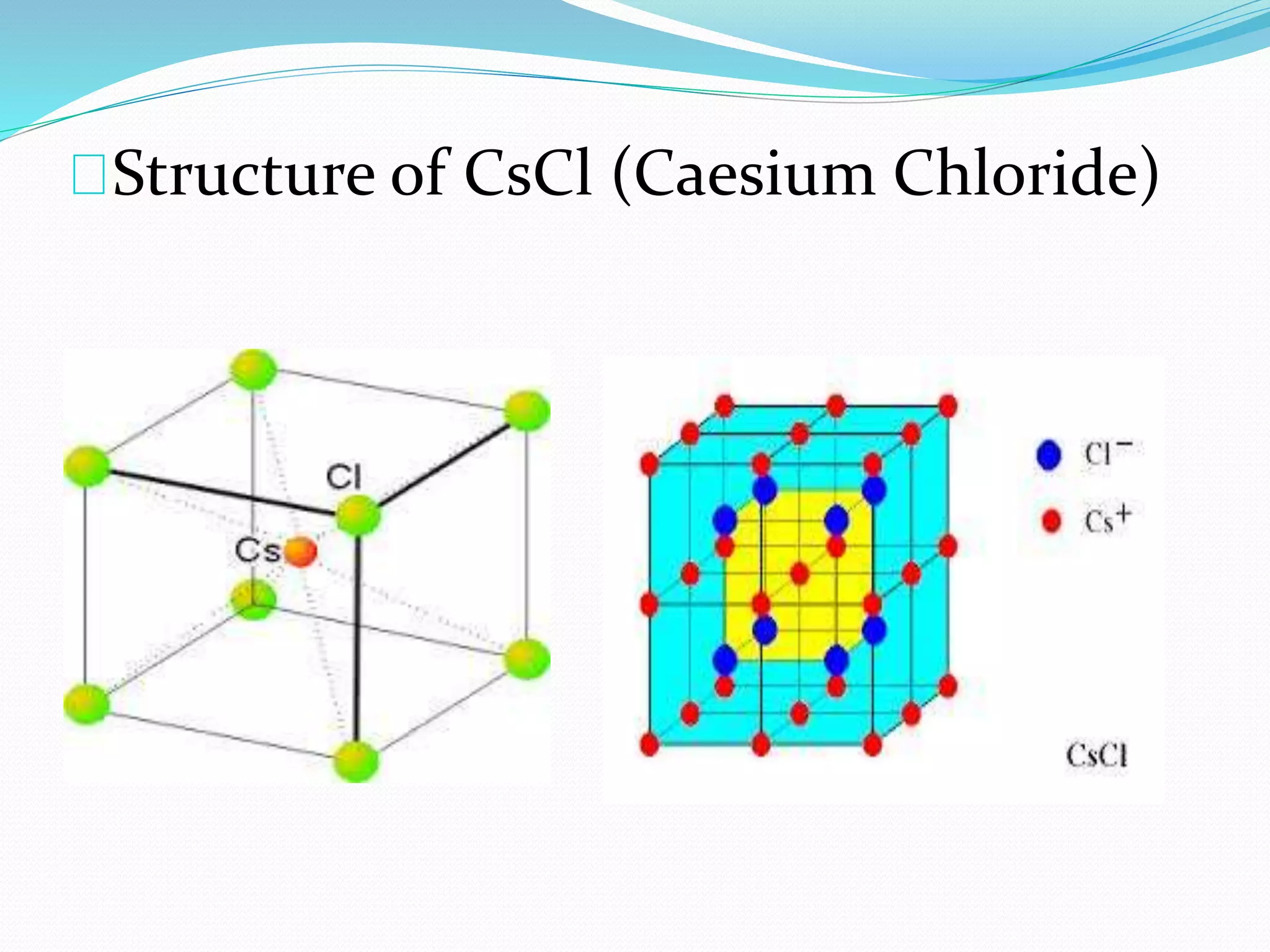
This document discusses different types of solids and their crystal structures. It describes crystalline solids as having long-range periodic atomic arrangements, while amorphous solids lack long-range order. Polycrystalline solids consist of many small crystallites. Common crystal structures include body-centered cubic, face-centered cubic, and hexagonal close-packed arrangements. Defects in crystal structures like point defects and dislocations are also summarized.















![Miller Indices: Equivalent Directions
z
y
Equivalent directions due to crystal symmetry:
x
1
2
3
1: [100]
2: [010]
3: [001]
Notation <100> used to denote all directions equivalent to [100]](https://image.slidesharecdn.com/solidstatechemistry-141012001414-conversion-gate01/75/Solid-state-chemistry-16-2048.jpg)

![z
y
Miller Indices of Planes
z=
x
y=
x=a
x y z
[1] Determine intercept of plane with each axis a ∞ ∞
[2] Invert the intercept values 1/a 1/∞ 1/∞
[3] Convert to the smallest integers 1 0 0
[4] Enclose the number in round brackets (1 0 0)](https://image.slidesharecdn.com/solidstatechemistry-141012001414-conversion-gate01/75/Solid-state-chemistry-18-2048.jpg)
![z
y
Miller Indices of Planes
x
x y z
[1] Determine intercept of plane with each axis 2a 2a 2a
[2] Invert the intercept values 1/2a 1/2a 1/2a
[3] Convert to the smallest integers 1 1 1
[4] Enclose the number in round brackets (1 1 1)](https://image.slidesharecdn.com/solidstatechemistry-141012001414-conversion-gate01/75/Solid-state-chemistry-19-2048.jpg)
![z
y
Planes with Negative Indices
x
x y z
[1] Determine intercept of plane with each axis a -a a
[2] Invert the intercept values 1/a -1/a 1/a
[3] Convert to the smallest integers 1 -1 1
[4] Enclose the number in round brackets 111](https://image.slidesharecdn.com/solidstatechemistry-141012001414-conversion-gate01/75/Solid-state-chemistry-20-2048.jpg)