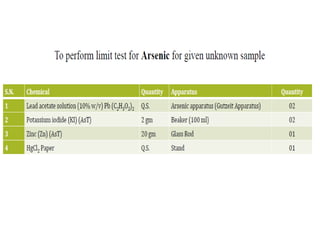
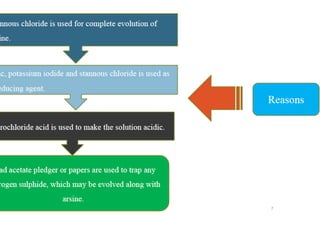
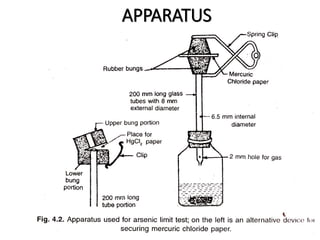
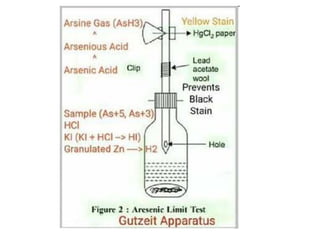
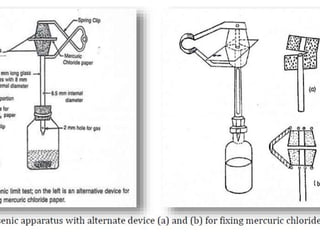
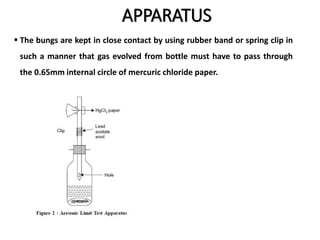
This document describes a test to detect the presence of arsenic in medicinal substances. The test works by reducing any arsenic in the sample to arsine gas using zinc, acid, and other reducing agents. The arsine gas is carried through a glass tube to mercuric chloride paper, where it will produce a yellow stain if arsenic is present. The intensity of the stain is compared to standard stains produced using known quantities of arsenic to determine if the level of arsenic passes the limit test. The test is run for 40 minutes and any yellow stain less intense than the standard passes, indicating the level of arsenic is below the limit.