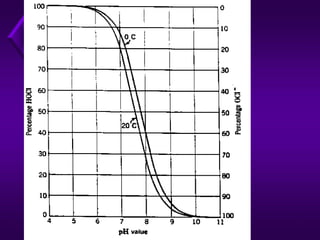
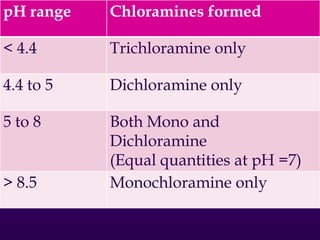
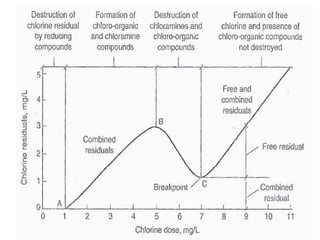
The document outlines various disinfection methods for drinking water, highlighting techniques such as boiling, chlorination, and UV irradiation. It discusses the chemistry behind chlorination, including the formation of hypochlorous acid and the significance of pH levels in disinfection effectiveness. The document also covers factors affecting chlorination efficiency and different applications like pre-chlorination and super chlorination.