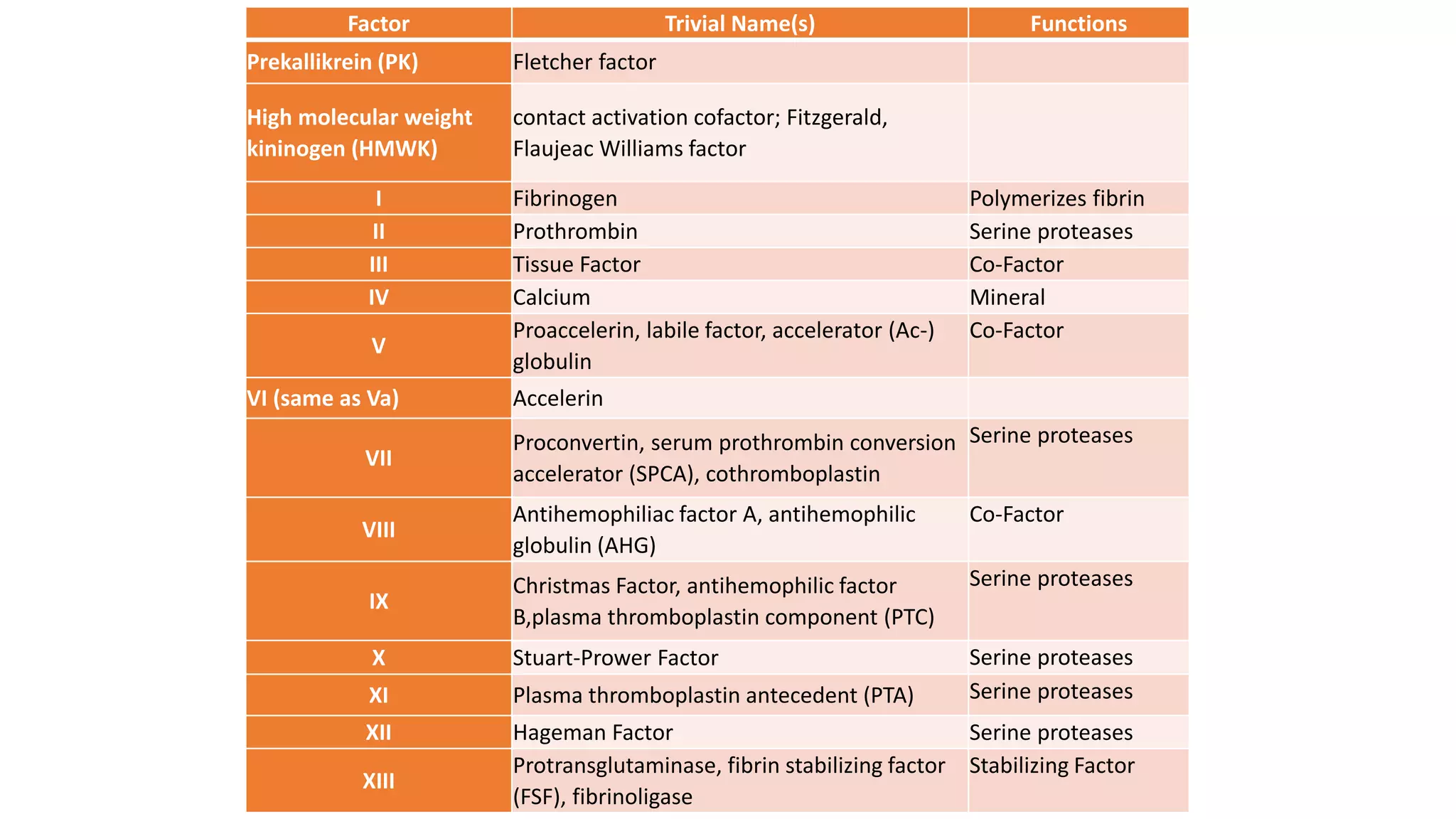
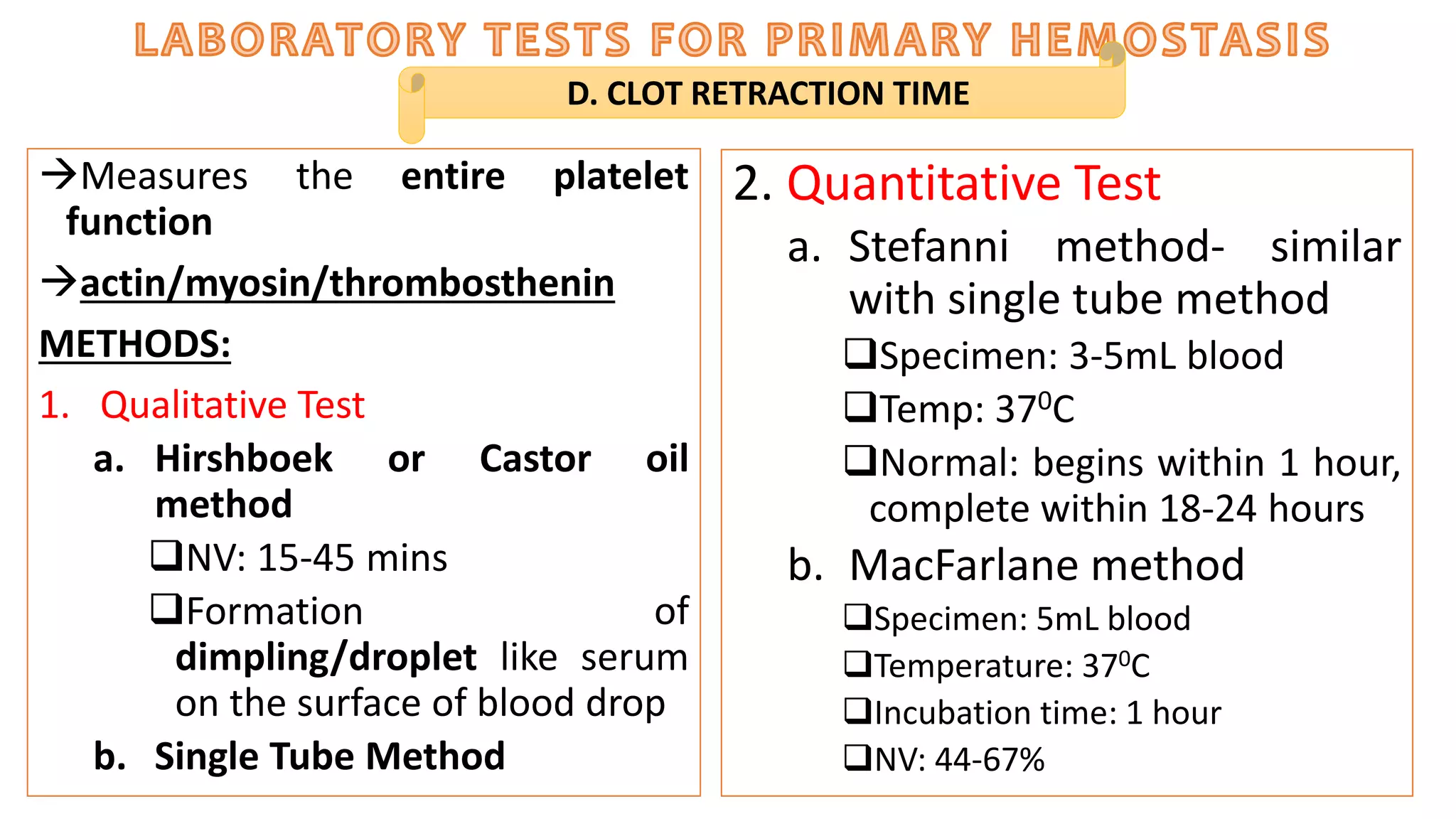
The document outlines the historical development and understanding of hemostasis and related disorders, particularly hemophilia, emphasizing key milestones from the 2nd century to the 20th century. It details the complex processes of primary and secondary hemostasis, the roles of various blood components, and the tests used to evaluate hemostatic mechanisms and platelet function. Additionally, it covers clinical manifestations of bleeding disorders and describes laboratory methods for platelet evaluation and bleeding time assessment.