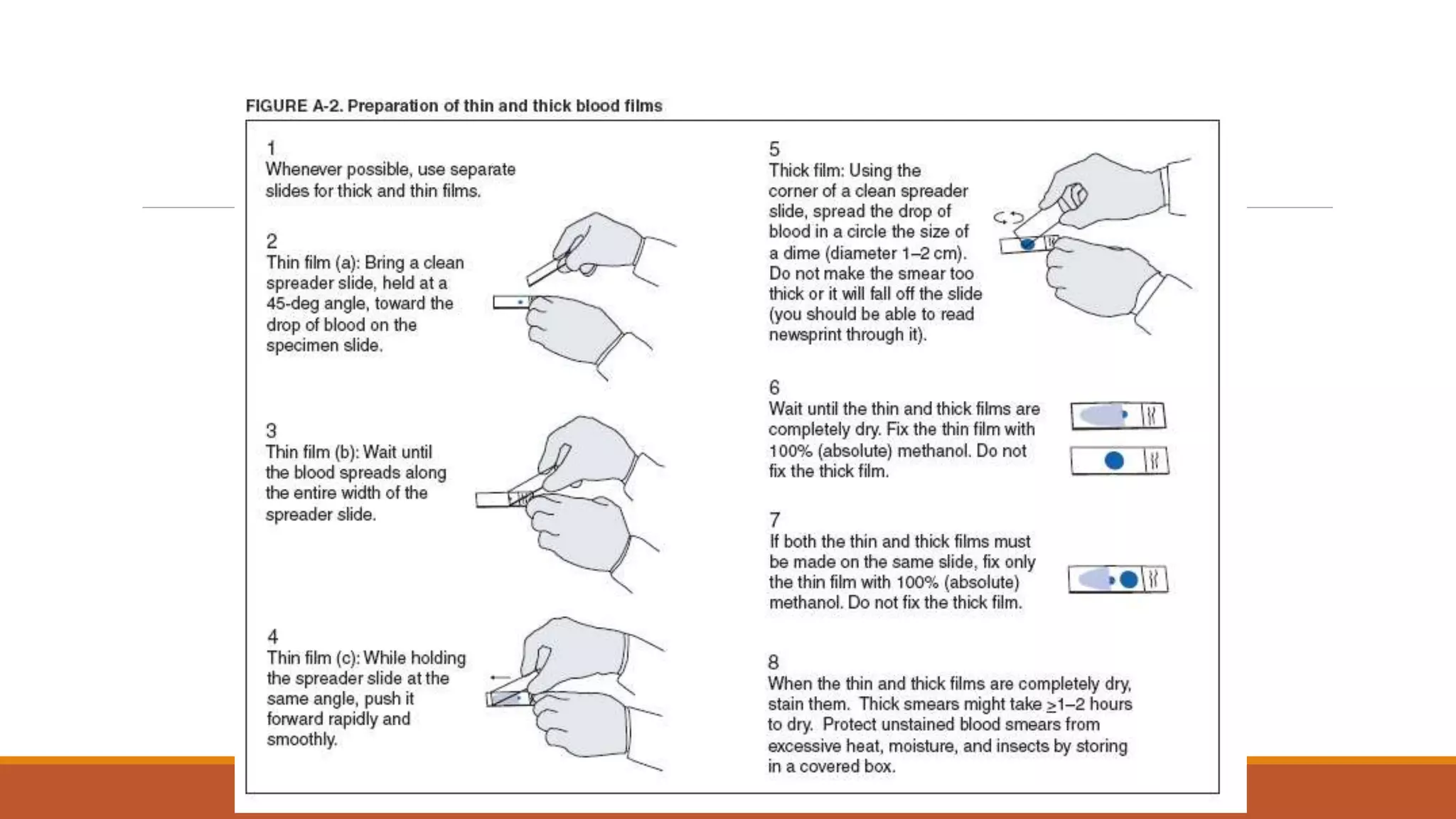
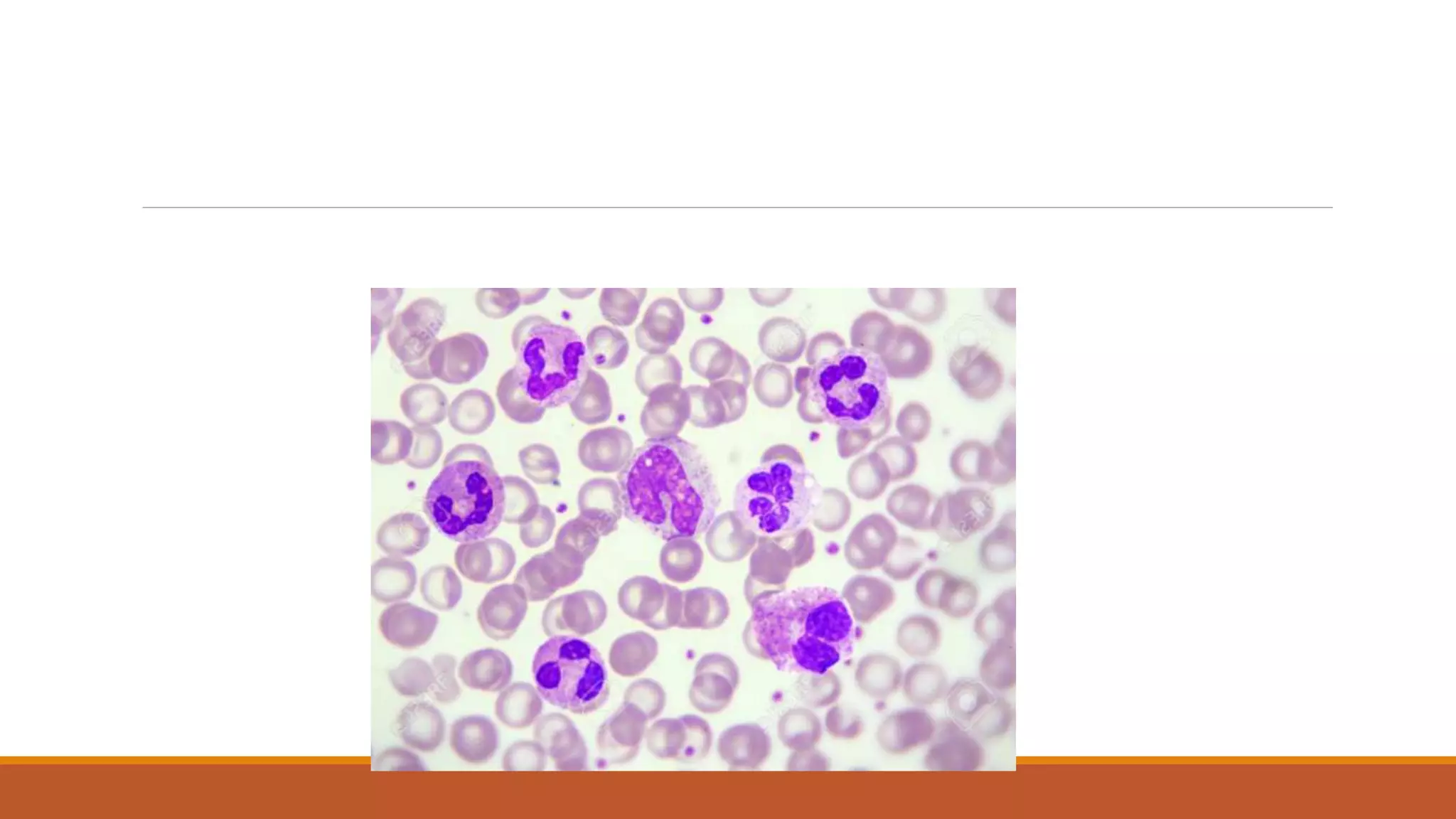
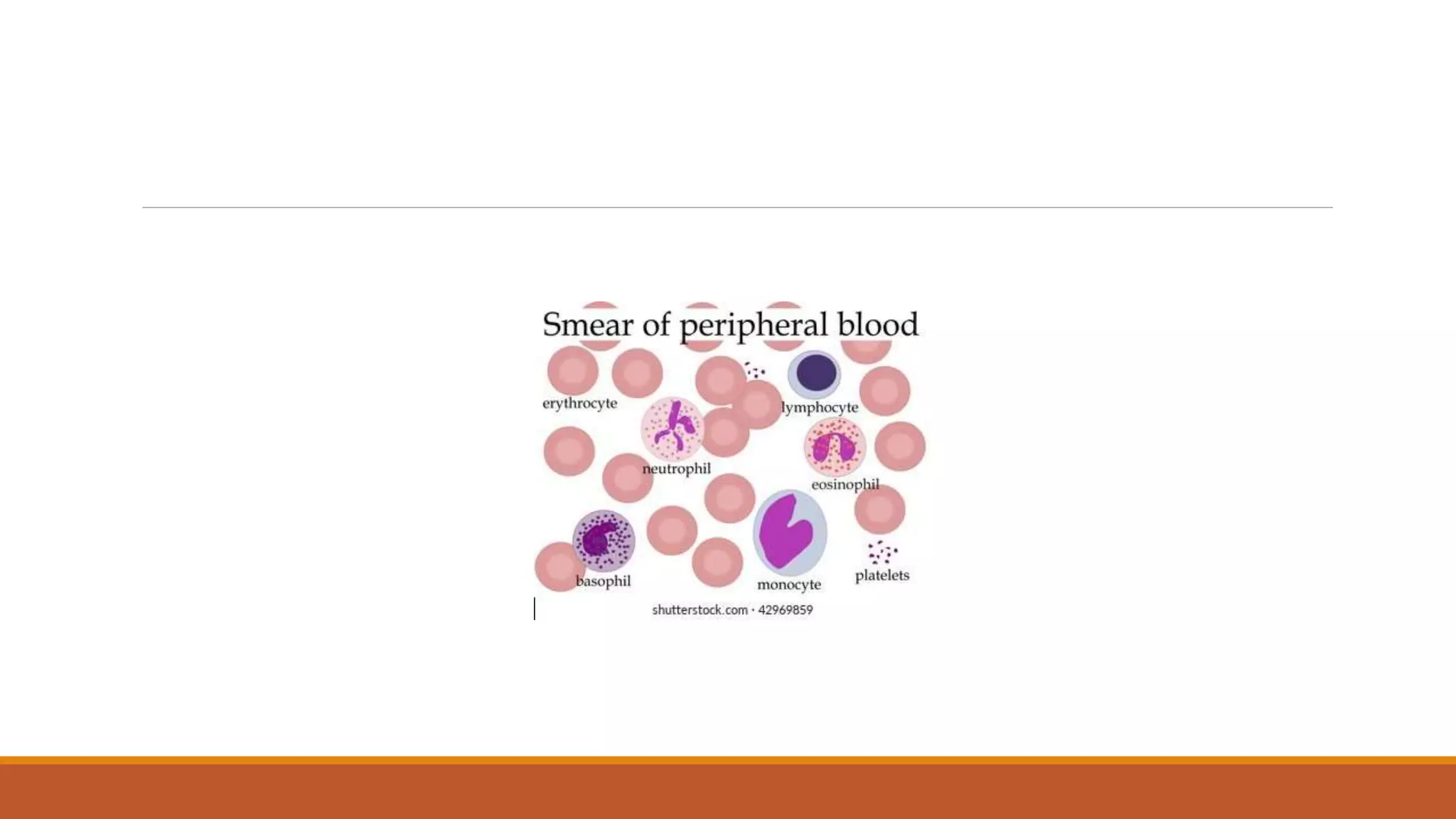
This document provides information on staining blood films and smears. It discusses the different types of stains used including Romanowsky stains like Leishman stain, Giemsa stain, Wright stain, and Field stain. Specimens should be collected in EDTA and smears prepared within an hour then fixed in methanol or ethanol to preserve cell morphology before staining. Romanowsky stains use methylene blue and eosin dyes to reveal subtle differences in cell structures and components.