Crystal defects occur when the regular patterns of atoms in crystalline materials are interrupted. There are several types of crystal defects including point defects, line defects, and plane defects. Point defects are defects that occur at or around a single lattice point and include vacancies, interstitials, and substitutions. Vacancies occur when an atom is missing from its normal position in the crystal lattice. Interstitials occur when an atom occupies a position between normal lattice sites. Substitutions occur when a foreign atom replaces a host atom in the lattice. The presence of defects is necessary for crystals to have stability at any non-zero temperature due to the contribution of defects to entropy.










![VACANCY CONCENTRATION IN A METAL
For low concentration of defects, a quantitative relation
between n,N,Ev and T may be obtained through the use
of Boltzmann statistics is justified since the lattice sites
are distinguishable although the metal ions occupying
them are not. Also, the interaction between defects is
neglected.
The total no. of different ways (W) in which a metal ion
can be removed is given by
W = N(N-1)(N-2)....(N-n+1)/n! = N!/(N-n)!n!
Further, the entropy change ( S) associated with the
process is equal to kB ln W,
Where kB is the Boltzmann constant.
Therefore,
S= kB ln[N!/(N-n)!n!] ……..Eq.(1)
The logarithmic factorial terms of Eq.(1) can be simplified
using the stirling’s approximation, which states that for
large values of x,
ln x! = x ln x-x](https://image.slidesharecdn.com/crystaldefects-141011234916-conversion-gate01/85/Crystal-defects-11-320.jpg)
![ Therefore ,
ln[N!/(N-n)!n!]
=ln N! – ln(N-n)!-ln n!
=N ln N-N-{(N-n) ln (N-n)-(N-n)}-n ln n + n
=N ln N- (N-n) ln (N-n)-n ln n
Now, the Helmholtz free energy change ( A) is given by
A = E – T S
= nEv – T S
Where E represents the total internal energy change .At
equilibrium,
(δ A/ δn)T =0
Now, δ/ δn[N ln N-(N-n ) ln (N-n)-n ln n]
= - δ/ δn{(N-n) ln (N-n)+ n ln n}
= -[δ{(N-n) ln (N-n)}/ δn + δ(n ln n)/ δn]
= -{(N-n)/(N-n)(-1)+ ln (N-n) x (-1)+n/n+ln n}](https://image.slidesharecdn.com/crystaldefects-141011234916-conversion-gate01/85/Crystal-defects-12-320.jpg)
![ = -{-1- ln (N-n) + 1 + ln n}
= ln (N-n/n)
Therefore, δ(T S)/ δn = kBT ln (N-n/n)
And, [δ( A)/ δn]T = 0 = Ev–kBT ln (N-n/n)
Where the partial differentiation is carried out with respect to the
variable ‘n’, since the total no. of lattice sites/metal atoms is a
constant.
Ev = kBT ln (N-n/n)
Or
(N-n/n)= exp(Ev/kBT)
If the no. of vacancies is much smaller than the total no. of atoms,
i.e., if n<<N,then
N-n=N
So that
n=N exp(-Ev/kBT) ……….eq.(2)
Usually, the no. of vacancies (n) is a very small fraction of the no.
of metal ions/atoms, so that Eq.(2) can be used to calculate the
number of fraction of vacancies so long as the temperature is far
removed from the melting point.](https://image.slidesharecdn.com/crystaldefects-141011234916-conversion-gate01/85/Crystal-defects-13-320.jpg)


![SCHOTTKY DEFECT CONCENTRATION IN AN IONIC CRYSTAL
Let ‘n’ be the number of Schottky defects produced by
removing nA+ cation and nB- anions, N the total no. of
cation-anion pairs and Es the average energy required
to create a Schottky defect. Then, the total no. of
different ways in which ‘n’ Schottky defects can be
produced will be given by
W = [N!/(N-n)!n!]2
The exponent 2 appears in the above relation
because there are n cations as well as n anions so that
the no. of different ways in which a cation or anion can
be removed is given by
N!/(N-n)!n!
And the total no. of different ways by the compound
probability, which is given by the product of the two.
Therefore,
S = 2kB ln [N!/(N-n)!n!]](https://image.slidesharecdn.com/crystaldefects-141011234916-conversion-gate01/85/Crystal-defects-16-320.jpg)
![Using Stirling’s approximation, we get
ln[N!/(N-n)!n! = N ln N-(N-n) ln (N-n)-n ln n
Further, the Helmholtz free energy change
A = E – T S
=nEs –2kBT{N ln N-(N-n)ln (N-n)-n ln n}
At equilibrium,
[δ A/ δn]T = 0 = Es – 2kBT ln [(N-n)/n]
Or
Es = 2kBT ln [(N-n)/n]
Or
[(N-n)]= exp (Es/2kBT)
If n<<N, then N-n=N so that
n = N exp (-Es/2kBT) ……….Eq(1)
Eq(1) is an adequate expression for the determination
of the no. of Schottky defects present in binary ionic
crystal such as NaCl and MgO at ordinary
temperatures, at which the interaction between defects
could be negligible since their no. is relatively small.](https://image.slidesharecdn.com/crystaldefects-141011234916-conversion-gate01/85/Crystal-defects-17-320.jpg)


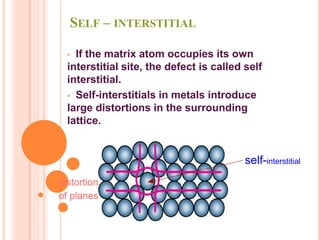
![Let Ei be the average energy required to displace an
atom from its regular lattice position to an interstitial,
Ni be the no. of interstitial sites and
N the total no. of atoms.
The no. of different ways in which ‘n’ interstitial
defects can be formed is given by,
W = N!/(N-n)!n! Ni!/(Ni-n)!n!
Therefore , the associated changes in entropy and
Helmholtz free energy will be,
S = kB ln [N!/(N-n)!n! Ni!/(Ni-n)!n!]
And
A = nEi – kBT ln [N!/(N-n)!n! Ni!/(Ni – n)!n!]](https://image.slidesharecdn.com/crystaldefects-141011234916-conversion-gate01/85/Crystal-defects-21-320.jpg)
![Using Stirling’s approximation, we get
ln [N!/(N-n)! n! Ni!/(Ni-n)! n!]
=ln N!/(N-n)! n! + ln Ni!/(Ni-n)! n!
= N ln n+ Ni ln N-(N-n) –(Ni-n) ln (Ni-n) – 2n ln n
At equilibrium,
[δ( A)/ δn]T = 0 = Ei – kBT ln {(N-n)(Ni-n)/n2}
Since the quantities N and Ni are independent of n .
Hence ,
(δN/ δn)T = 0 = (δNi/ δn)T
If N>>n and Ni>>n , then
Ei/kBT = ln (NNi/n2)
= ln (Nni) – 2 ln n
Therefore,
n = (NNi)1/2 exp ( - Ei/2kBT)](https://image.slidesharecdn.com/crystaldefects-141011234916-conversion-gate01/85/Crystal-defects-22-320.jpg)

![FRENKEL DEFECT CONCENTRATION
Let Ef be the average energy required to displace a
cation from its normal lattice position to an interstitial,
Ni be the no. of interstitial sites and
N the total no. of cation ( in a binary ionic crystal, A+
B-).
The no. of different ways in which ‘n’ Frenkel defects
can be produced is then given by,
Therefore, the associated changes in entropy and
Helmholtz free energy are given by,
And
W = N!/(N-n)!n! Ni!/(Ni-n)!n!
S = kB ln [N!/(N-n)!n! Ni!/(Ni-n)!n!]
A = nEf – kBT ln [N!/(N-n)!n! Ni!/(Ni – n)!n!]](https://image.slidesharecdn.com/crystaldefects-141011234916-conversion-gate01/85/Crystal-defects-24-320.jpg)
![The Stirlings formula gives,
ln [N!/(N-n)! n! Ni!/(Ni-n)! n!]
=ln N!/(N-n)! n! + ln Ni!/(Ni-n)! n!
= N ln N+ Ni ln Ni -(N-n) ln (N-n)–(Ni-n) ln (Ni-n) – 2n ln n
At equilibrium,
[δ( A)/ δn]T = 0 = Ef – kBT ln {(N-n)(Ni-n)/n2}
Since the terms containing constant quantities like N and Ni
disappear on differentiation.
If N>>n and Ni>>n, then
Ef / kBT = ln(NNi / n2)
= ln (NNi) – 2 ln n
Therefore,
n = (NNi)1/2 exp ( - Ef/2kBT)
Usually, the no. of frenkel defect is small at low
temperature.](https://image.slidesharecdn.com/crystaldefects-141011234916-conversion-gate01/85/Crystal-defects-25-320.jpg)