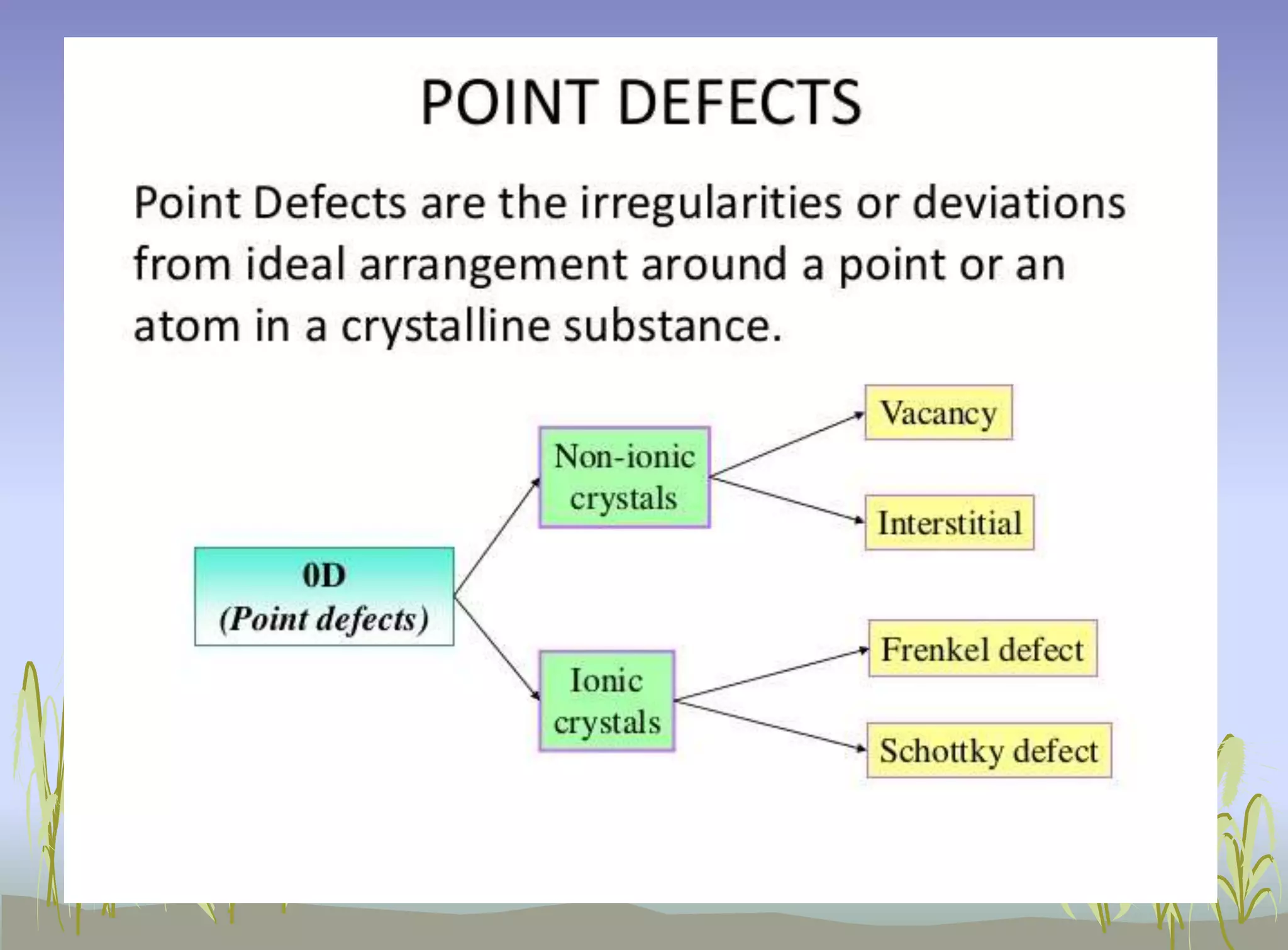
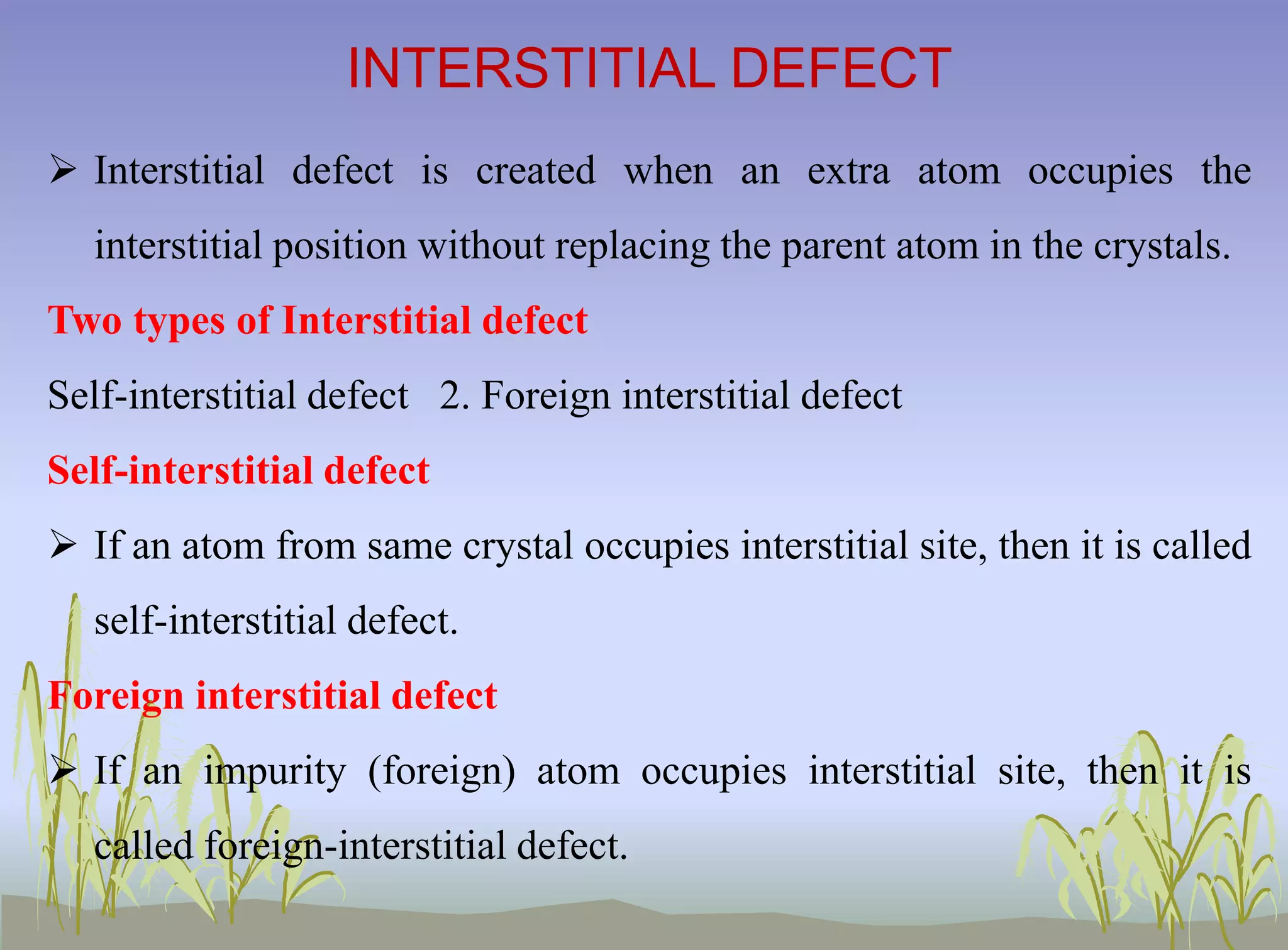
Crystal defects refer to any deviations from the regular geometric arrangement of atoms in a crystal structure. No crystal is truly perfect, as defects are always present due to imperfect packing during crystal formation and thermal vibrations. Common types of defects include vacancies where atomic sites are missing, interstitial defects where extra atoms occupy interstitial spaces, Schottky defects where an anion-cation pair is missing, and Frenkel defects where a cation shifts from its regular site to an interstitial site. Line defects called dislocations are also common, where the crystal structure is distorted along a line, and include edge dislocations from extra atomic planes and screw dislocations from spiral displacements of atoms. Defects significantly