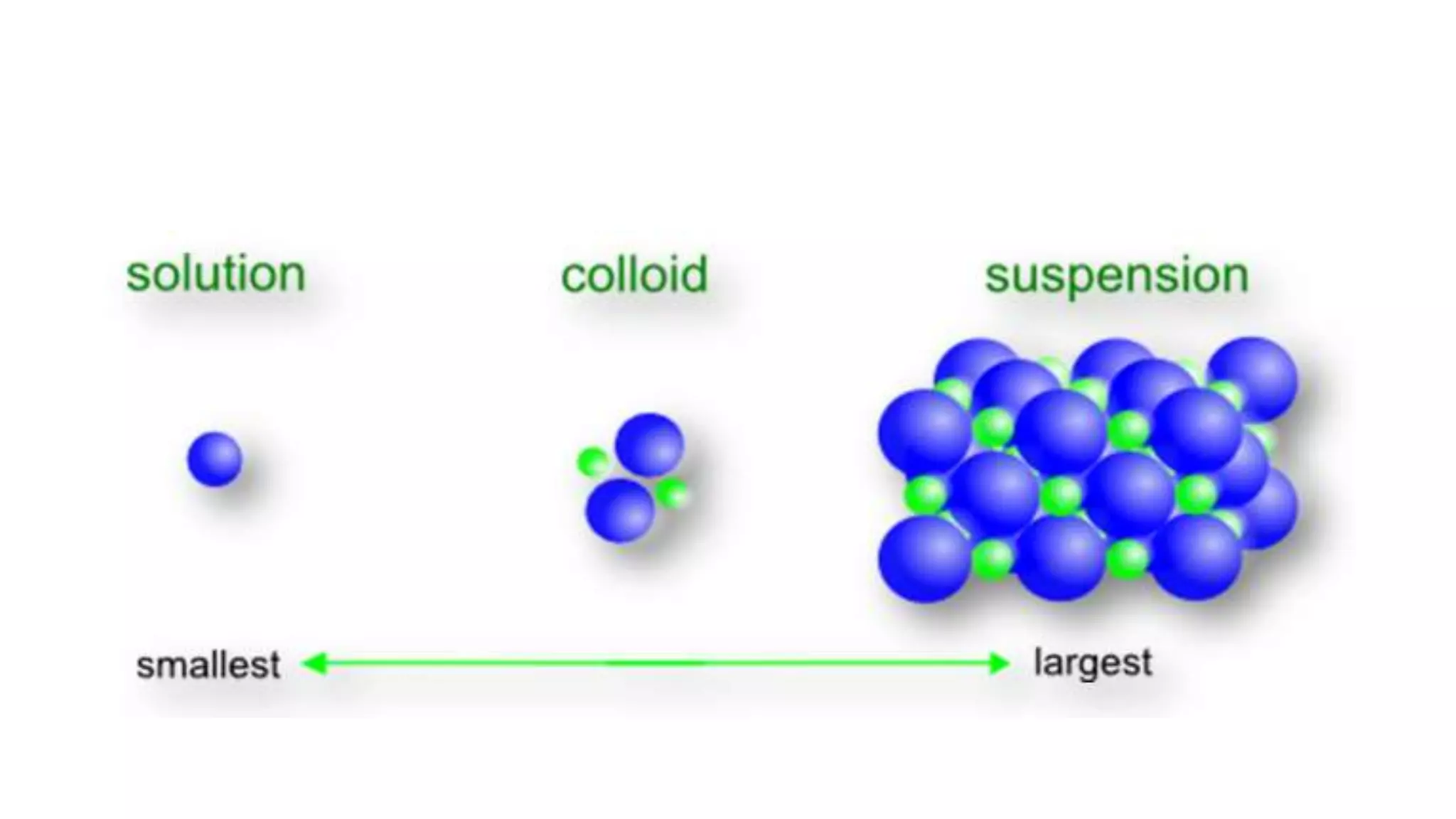
The document discusses colloids, defining them as a uniform dispersion of particles within a medium and detailing their applications in therapy, solubility enhancement, and targeted drug delivery. It contrasts colloidal and molecular dispersions based on properties such as particle size, visibility, and settling behavior. Furthermore, it covers characteristics of dispersed phases, including particle size, shape, surface area, and charge, as well as methods for purifying colloidal dispersions like dialysis and ultrafiltration.