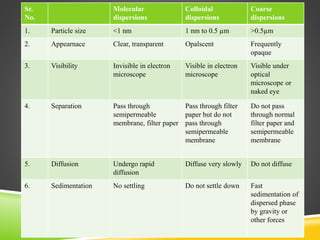
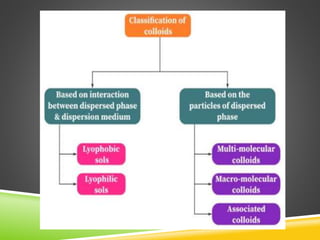
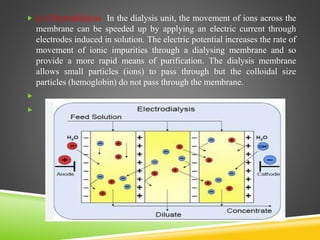
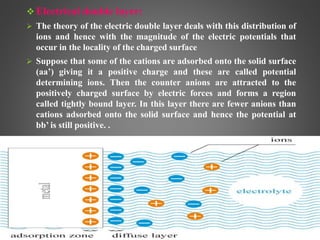
This document provides an overview of colloidal dispersions. It defines colloids as micro-heterogeneous dispersed systems with particle sizes between 1 nm and 1 μm. Colloids differ from molecular and coarse dispersions based on particle size, appearance, visibility, and ability to pass through membranes. Colloidal particles take on various shapes and their properties depend on shape. Colloids are classified as lyophilic or lyophobic based on particle-solvent interactions. The document discusses purification, association, optical, kinetic, electrical, and stability properties of colloids.