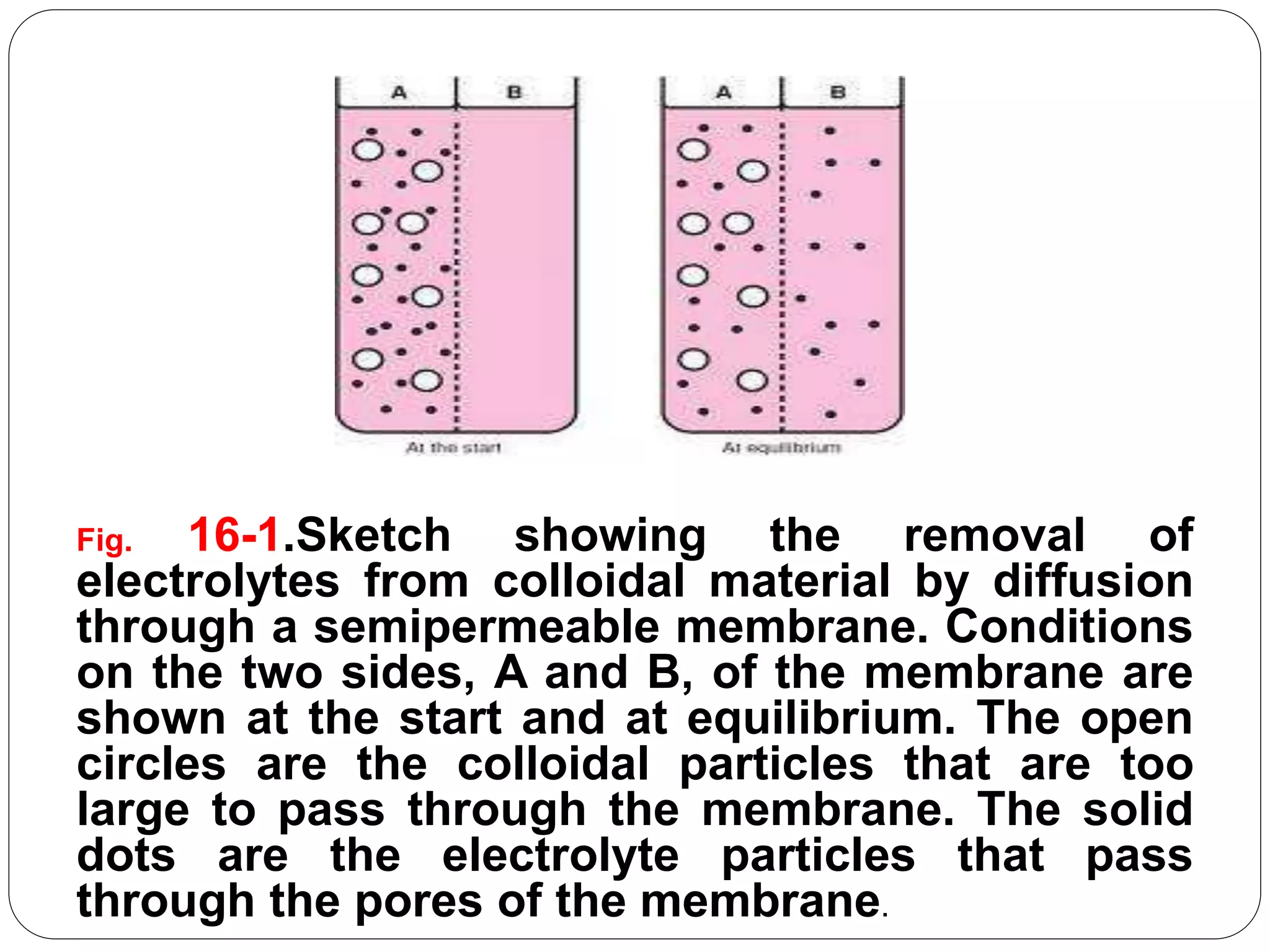
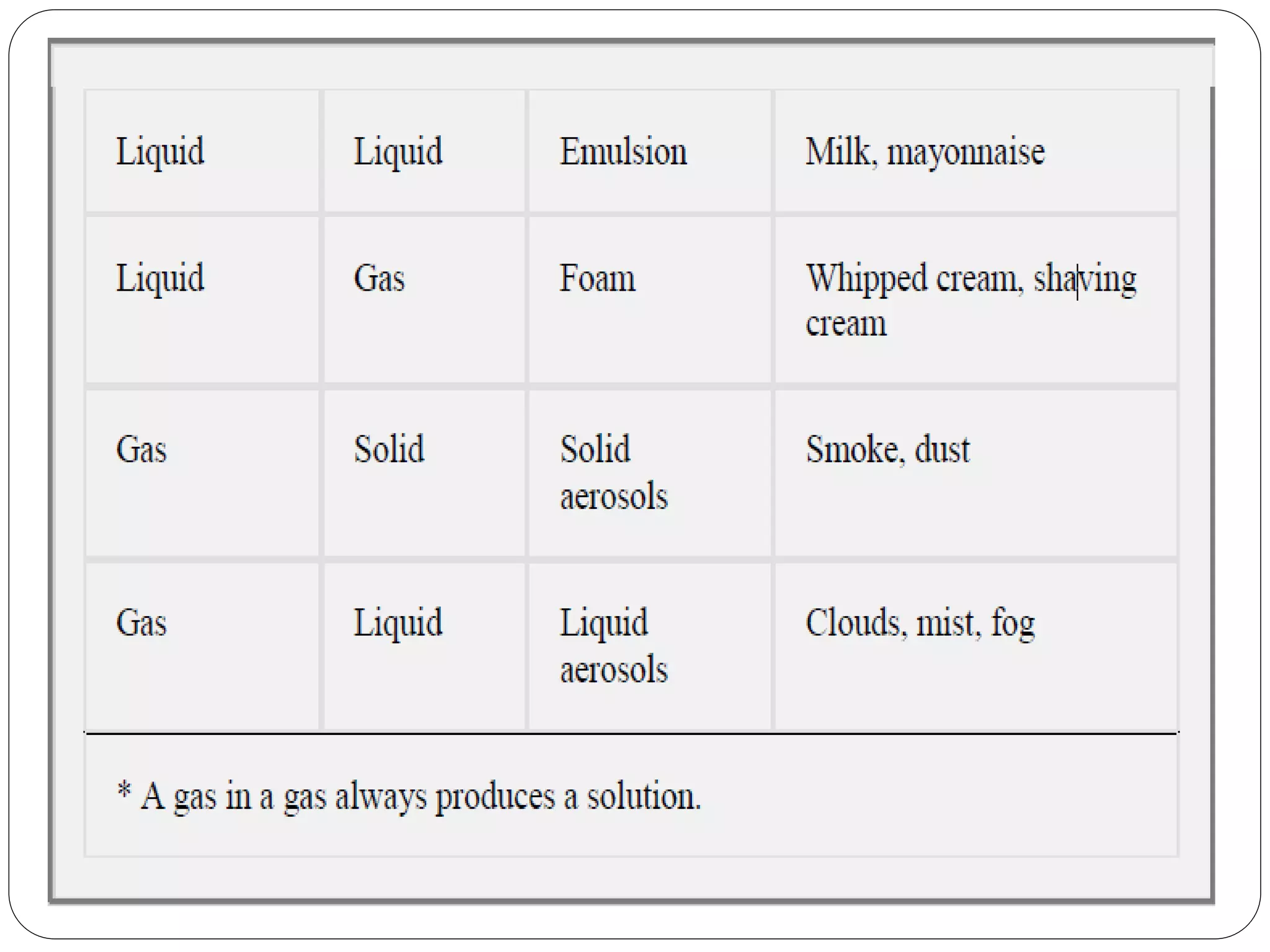
The document discusses colloidal dispersions, which are mixtures where particles of varying sizes are dispersed in a continuous medium. It classifies these systems into molecular, colloidal, and coarse dispersions, highlighting properties like particle size, interaction with solvents, and methods of separation. Additionally, it explains the unique characteristics of colloidal particles, such as their high specific surface area and their behavior under light scattering and diffusion.