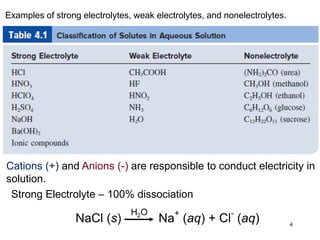
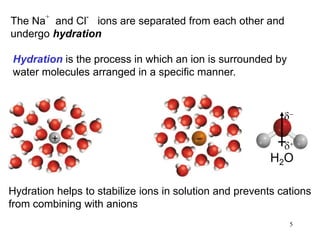
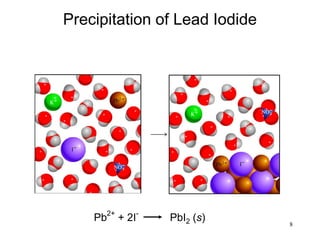
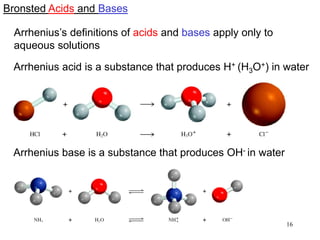
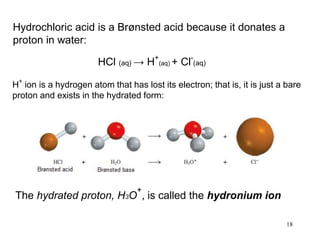
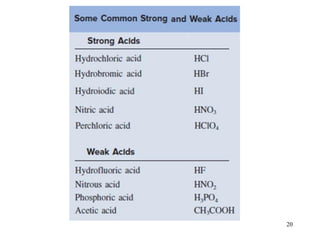
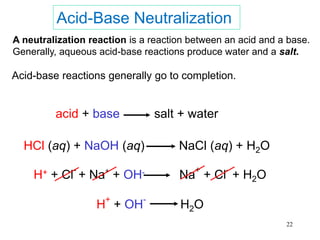
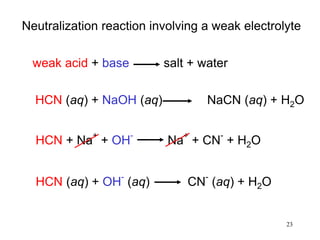
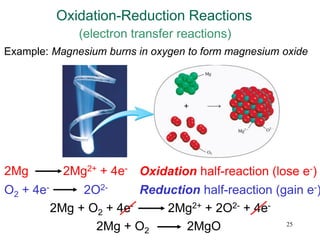
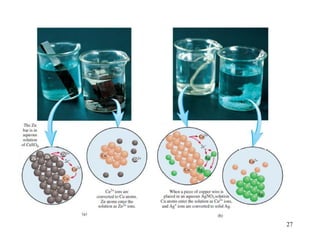
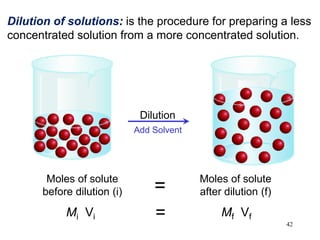
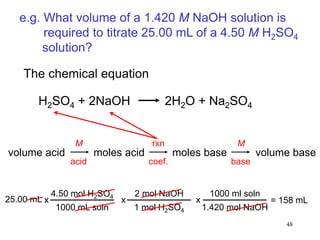
This document discusses various topics related to aqueous solutions and reactions. It begins by defining key terms like solute, solvent, electrolyte and providing examples. It then covers properties of aqueous solutions such as conductivity. Various acid-base reactions and concepts are explained like Brønsted-Lowry acids and bases, neutralization reactions. Oxidation-reduction reactions and oxidation numbers are also discussed. Finally, the document covers concentration of solutions and calculations involving molarity, dilution and preparation of solutions.