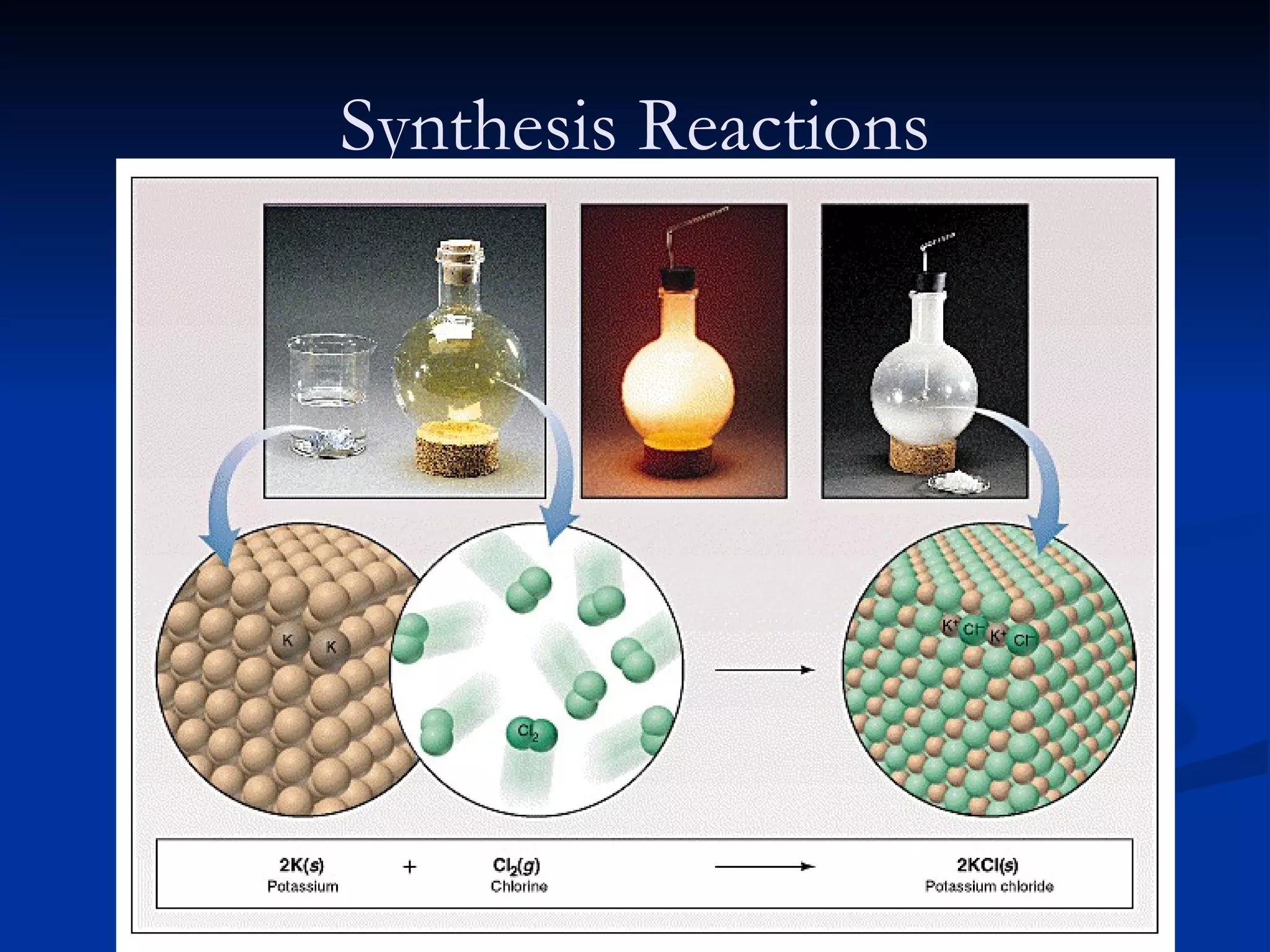
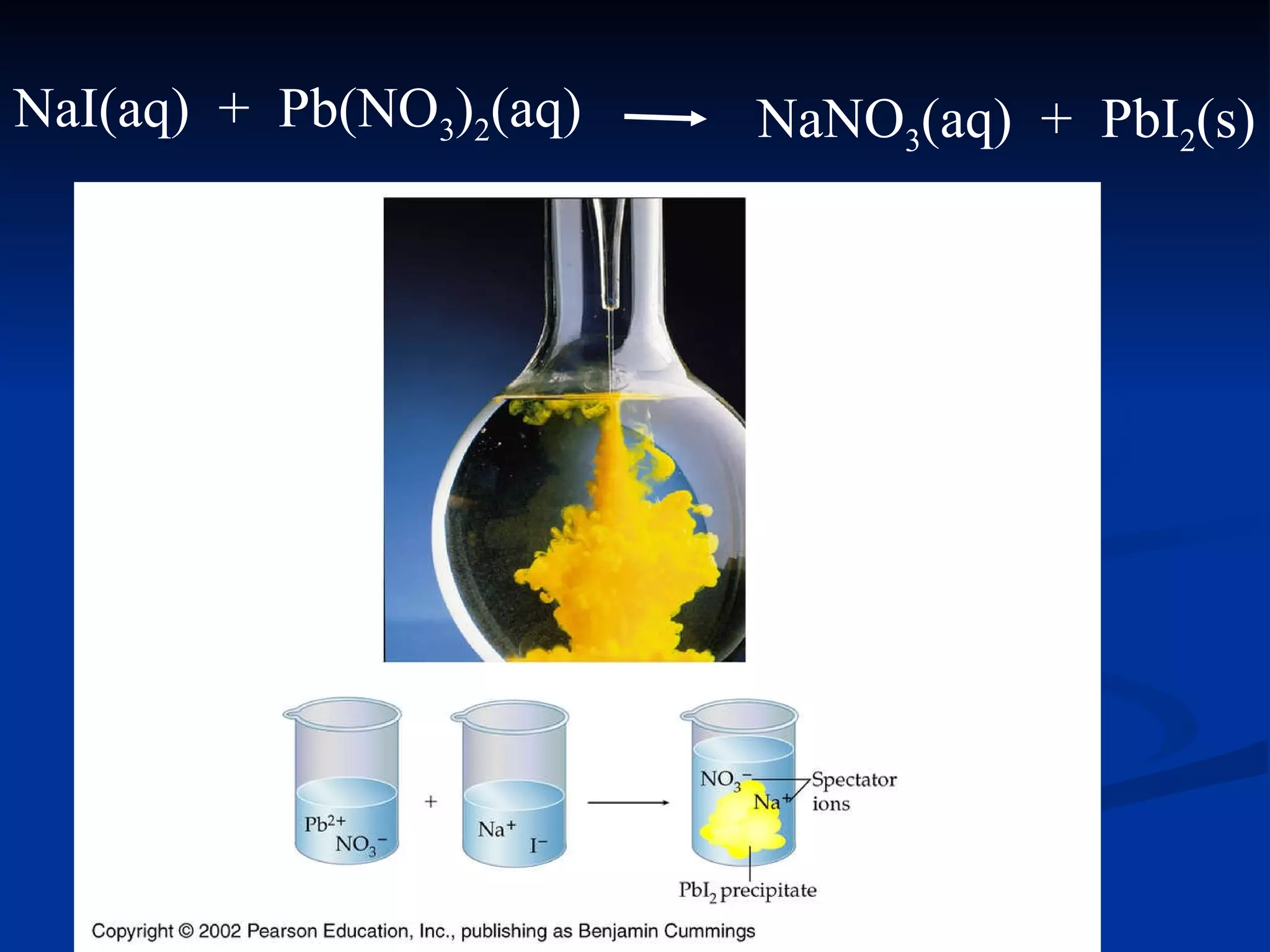
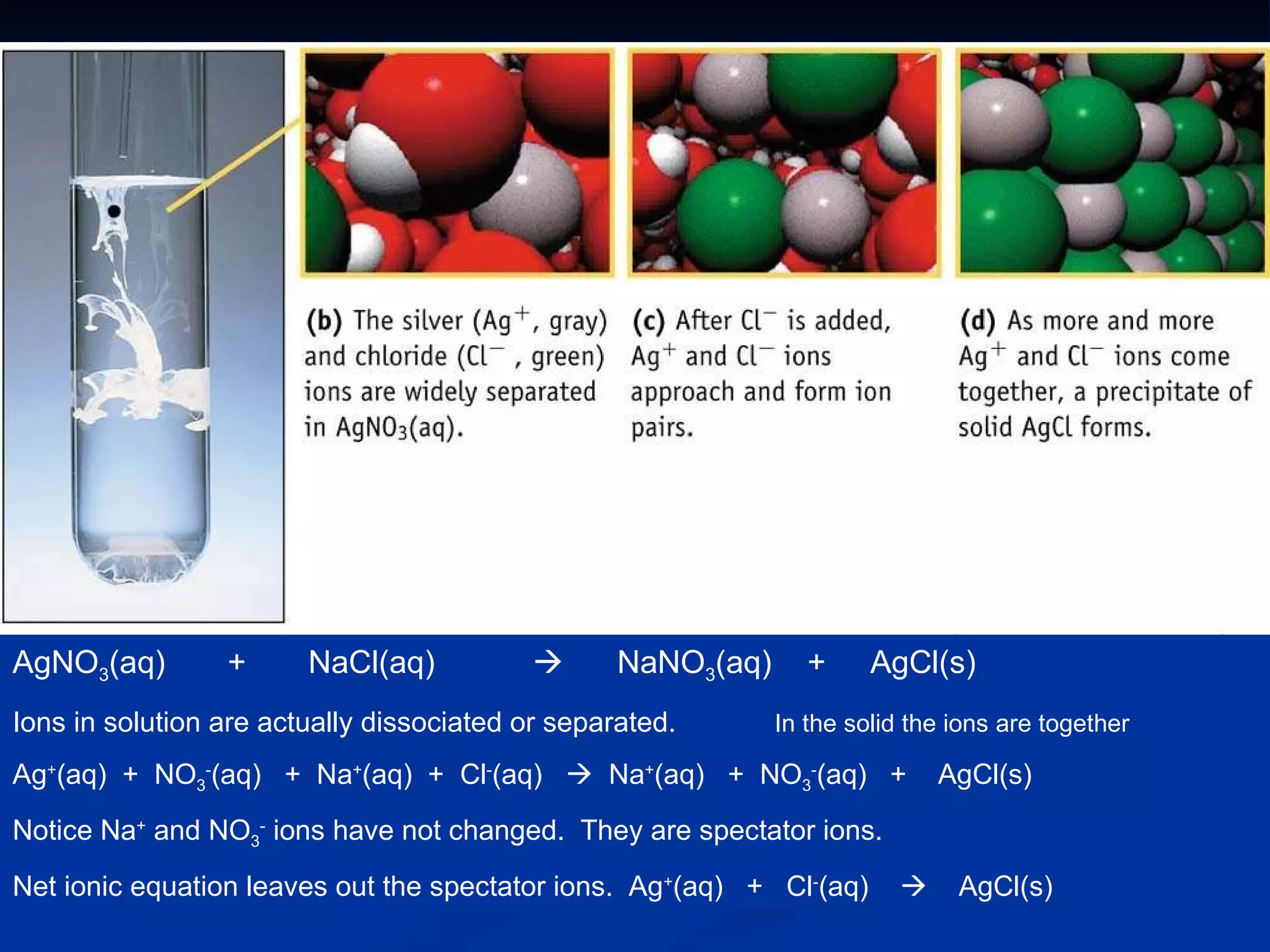
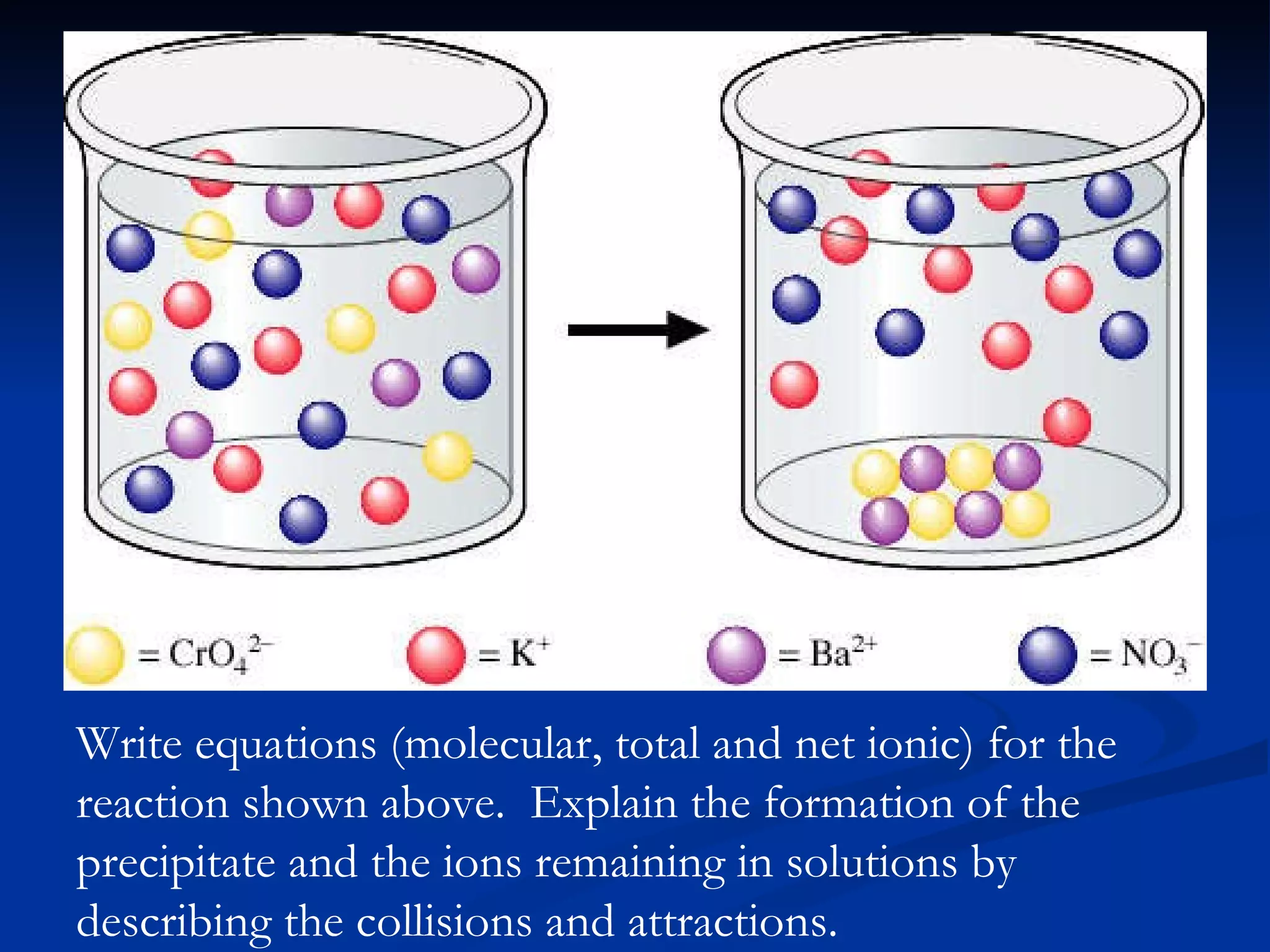
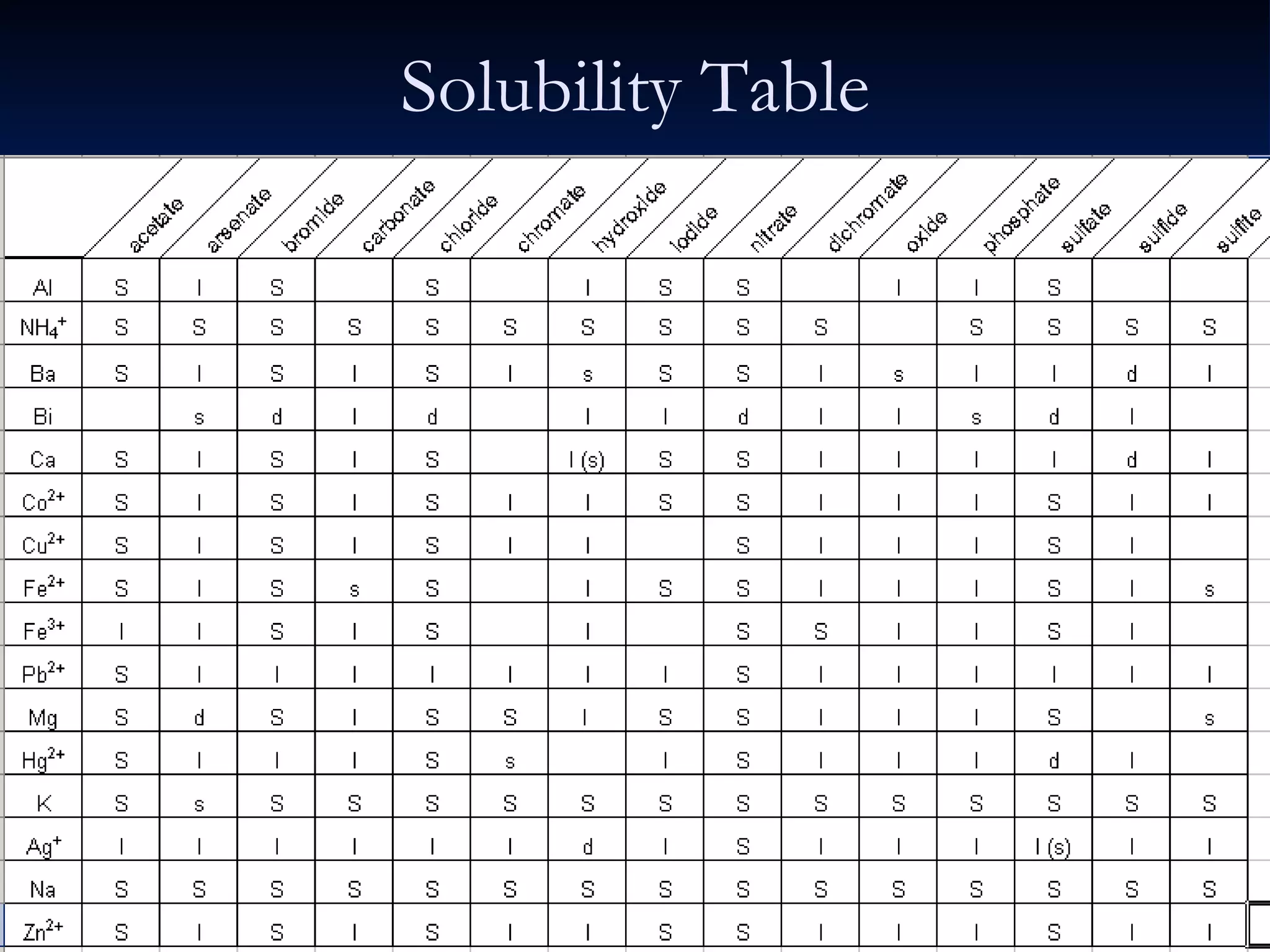
This document discusses different types of chemical reactions including decomposition, synthesis, combustion, double replacement, and single replacement reactions. It provides examples of each type of reaction and explains the key features that define them. Double replacement reactions are highlighted, where a metal replaces a metal in a compound and a nonmetal replaces a nonmetal, forming a precipitate if one of the products is insoluble. Guidelines are provided for writing molecular, total ionic, and net ionic equations for double replacement reactions.