04 Solutions
•Download as PPT, PDF•
0 likes•767 views
This document discusses different types of chemical reactions including precipitation, acid-base, and oxidation-reduction reactions. It provides examples of strong acids, weak acids, and bases. Neutralization reactions between acids and bases are described which produce water and a salt. Oxidation and reduction are defined as the loss or gain of electrons respectively. Half-reaction and oxidation state rules are outlined for balancing redox reactions.
Report
Share
Report
Share
Recommended
Lect w13 152_electrochemistry_key

The document provides information about a chemistry problem involving the molecular weight of a compound. A 3.41 x 10-6 g sample of the compound contains 4.67 x 10^16 molecules. Calculating the molecular weight based on the moles of molecules and mass of the sample gives a value of 44.0 g/mole. Of the compounds listed, CO2 has a molecular weight that matches this value.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
#18 Key

1. When ions dissolve in water, they are surrounded by water molecules (hydrated).
2. Precipitation will occur when Na2CO3 and AgNO3 or FeSO4 and Pb(NO3)2 are mixed, but not NaNO3 and NiSO4.
3. Acids donate H+ ions, bases accept H+ ions. A monoprotic acid has one ionizable H, a diprotic acid has two. Strong acids fully ionize, weak acids only partially ionize.
#12 Key

1. The document defines key chemistry terms including: aqueous solutions, solvents, solutes, electrolytes, nonelectrolytes, strong/weak electrolytes, and solvation.
2. Precipitation reactions are defined as reactions where an insoluble product called a precipitate forms. Molecular, complete ionic, spectator ions, and net ionic equations are also defined.
3. Strong acids and bases are listed as well as examples of soluble and insoluble compounds in water. Spectator ions are identified for sample precipitation reactions.
#13 Key

This document provides sample problems and questions for a general chemistry exam. It includes sample balanced equations for neutralization reactions and definitions of key terms like oxidation, reduction, concentration, and indicators. It also asks students to determine oxidation states, identify redox reactions, calculate molarity and amounts of substances in solutions, and determine concentrations of ions after mixing solutions.
Balancing redox reactions 

Redox reactions involve the transfer of electrons between species. There are two types of agents involved - oxidizing agents that reduce other species by accepting electrons, and reducing agents that oxidize other species by donating electrons. Identification of redox reactions involves looking for a change in oxidation state between reactants and products. Balancing redox reactions uses the ion-electron method of writing and balancing half reactions for oxidation and reduction and combining them. Organic redox reactions use a similar process by writing oxidation and reduction half reactions and balancing mass, charge, and electrons.
#17 Key

This document provides sample chemistry problems and questions related to topics like:
- Writing balanced equations for neutralization reactions
- Defining terms like oxidation, reduction, concentration, and indicators
- Determining oxidation states of elements in compounds
- Identifying redox, precipitation, and acid-base reactions
- Performing calculations involving molarity, moles, and concentration
The problems cover a wide range of general chemistry concepts.
Chem Unit5

The document discusses various chemistry concepts including:
1. Avogadro's number defines the number of particles in one mole of a substance as 6.02x1023.
2. Molar mass is the mass of one mole of a pure substance and is used to convert between grams and moles.
3. Empirical and molecular formulas can be determined from percent composition data using mole ratios and molar mass.
4. Chemical equations represent chemical reactions and must be balanced to satisfy the law of conservation of mass.
Recommended
Lect w13 152_electrochemistry_key

The document provides information about a chemistry problem involving the molecular weight of a compound. A 3.41 x 10-6 g sample of the compound contains 4.67 x 10^16 molecules. Calculating the molecular weight based on the moles of molecules and mass of the sample gives a value of 44.0 g/mole. Of the compounds listed, CO2 has a molecular weight that matches this value.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
#18 Key

1. When ions dissolve in water, they are surrounded by water molecules (hydrated).
2. Precipitation will occur when Na2CO3 and AgNO3 or FeSO4 and Pb(NO3)2 are mixed, but not NaNO3 and NiSO4.
3. Acids donate H+ ions, bases accept H+ ions. A monoprotic acid has one ionizable H, a diprotic acid has two. Strong acids fully ionize, weak acids only partially ionize.
#12 Key

1. The document defines key chemistry terms including: aqueous solutions, solvents, solutes, electrolytes, nonelectrolytes, strong/weak electrolytes, and solvation.
2. Precipitation reactions are defined as reactions where an insoluble product called a precipitate forms. Molecular, complete ionic, spectator ions, and net ionic equations are also defined.
3. Strong acids and bases are listed as well as examples of soluble and insoluble compounds in water. Spectator ions are identified for sample precipitation reactions.
#13 Key

This document provides sample problems and questions for a general chemistry exam. It includes sample balanced equations for neutralization reactions and definitions of key terms like oxidation, reduction, concentration, and indicators. It also asks students to determine oxidation states, identify redox reactions, calculate molarity and amounts of substances in solutions, and determine concentrations of ions after mixing solutions.
Balancing redox reactions 

Redox reactions involve the transfer of electrons between species. There are two types of agents involved - oxidizing agents that reduce other species by accepting electrons, and reducing agents that oxidize other species by donating electrons. Identification of redox reactions involves looking for a change in oxidation state between reactants and products. Balancing redox reactions uses the ion-electron method of writing and balancing half reactions for oxidation and reduction and combining them. Organic redox reactions use a similar process by writing oxidation and reduction half reactions and balancing mass, charge, and electrons.
#17 Key

This document provides sample chemistry problems and questions related to topics like:
- Writing balanced equations for neutralization reactions
- Defining terms like oxidation, reduction, concentration, and indicators
- Determining oxidation states of elements in compounds
- Identifying redox, precipitation, and acid-base reactions
- Performing calculations involving molarity, moles, and concentration
The problems cover a wide range of general chemistry concepts.
Chem Unit5

The document discusses various chemistry concepts including:
1. Avogadro's number defines the number of particles in one mole of a substance as 6.02x1023.
2. Molar mass is the mass of one mole of a pure substance and is used to convert between grams and moles.
3. Empirical and molecular formulas can be determined from percent composition data using mole ratios and molar mass.
4. Chemical equations represent chemical reactions and must be balanced to satisfy the law of conservation of mass.
Chapter 19.2 : Balancing Redox Equations

1. The half-reaction method consists of 7 steps to balance redox equations. This includes writing oxidation and reduction half-reactions, balancing atoms and charge, and combining the half-reactions.
2. The reaction of potassium permanganate with iron(II) sulfate is used as an example. The oxidation half-reaction involves manganese and the reduction half-reaction involves iron.
3. The balanced equation for the reaction is: 10FeSO4 + 2KMnO4 + 8H2SO4 → 5Fe3(SO4)2 + 2MnSO4 + K2SO4 + 8H2O.
04

The document discusses various topics related to reactions in aqueous solutions including:
- Solutions, solvents, solutes, electrolytes, and nonelectrolytes
- Strong and weak electrolytes and their ionization
- Acid-base theories and classifications of acids and bases
- Oxidation-reduction reactions and oxidation numbers
- Precipitation, acid-base, and redox reactions
- Solution stoichiometry, dilution, titrations, and gravimetric analysis
Elektrolit dan Nonelek

This document provides an overview of chapter 4 from a chemistry textbook, which covers chemical reactions. It begins by defining electrolytes, non-electrolytes, and discussing the properties of aqueous solutions. It then covers the three main types of reactions that occur in aqueous solutions: precipitation reactions, acid-base reactions involving proton transfer, and redox reactions involving electron transfer. Specific examples of each reaction type are provided. Key concepts around oxidation, reduction, and oxidation numbers are also explained.
Tang 02 balancing redox reactions 2

The document discusses balancing redox reactions using the half-reaction method. It provides several examples of writing and balancing half-reactions and using them to derive the overall balanced redox equation. Key steps include separating the reaction into oxidation and reduction half-reactions, balancing all elements except H and O, adding H2O to balance O, adding H+ or OH- to balance H, and adding electrons to balance charge.
C20 Review Unit 02 Chemical Reactions

1) A chemical reaction can be differentiated from a phase change or nuclear change based on whether it involves a change in chemical composition, state of matter, or type of element.
2) In a chemical reaction, reactants yield products through the rearrangement of atoms. Chemical equations use symbols to represent the reactants and products.
3) Evidence that a chemical reaction occurred includes the release or absorption of energy, production of a gas, formation of a precipitate, or a color change.
#13

The document contains questions about chemistry concepts including:
1) Writing balanced molecular and net ionic equations for neutralization reactions between acids and bases.
2) Defining oxidation, reduction, concentration, molarity, and indicators.
3) Asking if oxidation can occur without accompanying reduction.
4) Calculating oxidation numbers of elements in compounds.
5) Identifying redox, precipitation, and acid-base reactions based on balanced equations.
6) Calculating molarity, moles of solute, and volumes of solutions.
#10 Key

This document contains 7 problems related to general chemistry. It provides chemical equations to balance, asks students to calculate formula weights and percent compositions of substances, defines key concepts like moles and molar mass, and has multi-step calculations involving moles, molar mass, and grams. The problems cover topics like stoichiometry, formula weights, percent composition, gas laws, and molar conversions.
Chemistry formula list 1 (Examville.com)

The document provides information on chemical formulae, equations, calculations involving moles and molar mass/volume. It also covers the chemical properties and reactions of group 1 and 17 elements, as well as properties of salts such as solubility, color, and the effects of heating on different salts such as carbonates and nitrates.
Lect w13 152_electrochemistry_abbrev

The document discusses an electrochemistry unit covering chemical reactions that produce electrical currents or voltages. It provides information on voltaic cells, also known as galvanic cells, which harness spontaneous redox reactions to generate electricity. The document explains that voltaic cells use two half-reactions, an oxidation reaction at the anode and a reduction reaction at the cathode, to drive electrons from the anode to the cathode through an external circuit. Standard reduction potentials are used to predict if reactions will occur spontaneously.
Balancing equationsedit-091221120616-phpapp01

The document discusses techniques for writing and balancing chemical equations, including representing molecules with chemical formulas, determining the number of atoms in different compounds, using conservation of mass to balance equations, and examples of balancing a variety of chemical reactions. It provides guidance on balancing a number of sample equations involving different elements and compounds.
Properties of Solution

1. The document discusses the properties of solutions including solutes, solvents, electrolytes, and non-electrolytes. Solutions are homogeneous mixtures of substances where one substance is in smaller amounts (solute) and the other is the continuous liquid medium (solvent).
2. The document also covers acid-base reactions and redox reactions that occur in aqueous solutions including precipitation, acid-base, and redox reactions. Precipitation reactions form insoluble products. Acid-base reactions involve proton transfer and redox reactions involve electron transfer.
3. Oxidation states and oxidation numbers are discussed as well as rules for determining oxidation numbers of elements in compounds. Redox reactions involve oxidation, where electrons
Chapter 8: Reactions in Aqueous Solution

This chapter discusses different types of chemical reactions in aqueous solutions. It introduces driving forces that cause reactions, such as formation of a solid, water, or gas. It explains how to predict products using solubility rules and oxidation-reduction reactions when metals react with nonmetals. Reactions are classified into double displacement, acid-base, single replacement, combustion, synthesis, or decomposition reactions based on their driving forces.
Ch4 Reactions in Aqueous Solution

This document discusses various topics related to aqueous solutions and reactions. It begins by defining key terms like solute, solvent, electrolyte and providing examples. It then covers properties of aqueous solutions such as conductivity. Various acid-base reactions and concepts are explained like Brønsted-Lowry acids and bases, neutralization reactions. Oxidation-reduction reactions and oxidation numbers are also discussed. Finally, the document covers concentration of solutions and calculations involving molarity, dilution and preparation of solutions.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
More Related Content
What's hot
Chapter 19.2 : Balancing Redox Equations

1. The half-reaction method consists of 7 steps to balance redox equations. This includes writing oxidation and reduction half-reactions, balancing atoms and charge, and combining the half-reactions.
2. The reaction of potassium permanganate with iron(II) sulfate is used as an example. The oxidation half-reaction involves manganese and the reduction half-reaction involves iron.
3. The balanced equation for the reaction is: 10FeSO4 + 2KMnO4 + 8H2SO4 → 5Fe3(SO4)2 + 2MnSO4 + K2SO4 + 8H2O.
04

The document discusses various topics related to reactions in aqueous solutions including:
- Solutions, solvents, solutes, electrolytes, and nonelectrolytes
- Strong and weak electrolytes and their ionization
- Acid-base theories and classifications of acids and bases
- Oxidation-reduction reactions and oxidation numbers
- Precipitation, acid-base, and redox reactions
- Solution stoichiometry, dilution, titrations, and gravimetric analysis
Elektrolit dan Nonelek

This document provides an overview of chapter 4 from a chemistry textbook, which covers chemical reactions. It begins by defining electrolytes, non-electrolytes, and discussing the properties of aqueous solutions. It then covers the three main types of reactions that occur in aqueous solutions: precipitation reactions, acid-base reactions involving proton transfer, and redox reactions involving electron transfer. Specific examples of each reaction type are provided. Key concepts around oxidation, reduction, and oxidation numbers are also explained.
Tang 02 balancing redox reactions 2

The document discusses balancing redox reactions using the half-reaction method. It provides several examples of writing and balancing half-reactions and using them to derive the overall balanced redox equation. Key steps include separating the reaction into oxidation and reduction half-reactions, balancing all elements except H and O, adding H2O to balance O, adding H+ or OH- to balance H, and adding electrons to balance charge.
C20 Review Unit 02 Chemical Reactions

1) A chemical reaction can be differentiated from a phase change or nuclear change based on whether it involves a change in chemical composition, state of matter, or type of element.
2) In a chemical reaction, reactants yield products through the rearrangement of atoms. Chemical equations use symbols to represent the reactants and products.
3) Evidence that a chemical reaction occurred includes the release or absorption of energy, production of a gas, formation of a precipitate, or a color change.
#13

The document contains questions about chemistry concepts including:
1) Writing balanced molecular and net ionic equations for neutralization reactions between acids and bases.
2) Defining oxidation, reduction, concentration, molarity, and indicators.
3) Asking if oxidation can occur without accompanying reduction.
4) Calculating oxidation numbers of elements in compounds.
5) Identifying redox, precipitation, and acid-base reactions based on balanced equations.
6) Calculating molarity, moles of solute, and volumes of solutions.
#10 Key

This document contains 7 problems related to general chemistry. It provides chemical equations to balance, asks students to calculate formula weights and percent compositions of substances, defines key concepts like moles and molar mass, and has multi-step calculations involving moles, molar mass, and grams. The problems cover topics like stoichiometry, formula weights, percent composition, gas laws, and molar conversions.
Chemistry formula list 1 (Examville.com)

The document provides information on chemical formulae, equations, calculations involving moles and molar mass/volume. It also covers the chemical properties and reactions of group 1 and 17 elements, as well as properties of salts such as solubility, color, and the effects of heating on different salts such as carbonates and nitrates.
Lect w13 152_electrochemistry_abbrev

The document discusses an electrochemistry unit covering chemical reactions that produce electrical currents or voltages. It provides information on voltaic cells, also known as galvanic cells, which harness spontaneous redox reactions to generate electricity. The document explains that voltaic cells use two half-reactions, an oxidation reaction at the anode and a reduction reaction at the cathode, to drive electrons from the anode to the cathode through an external circuit. Standard reduction potentials are used to predict if reactions will occur spontaneously.
Balancing equationsedit-091221120616-phpapp01

The document discusses techniques for writing and balancing chemical equations, including representing molecules with chemical formulas, determining the number of atoms in different compounds, using conservation of mass to balance equations, and examples of balancing a variety of chemical reactions. It provides guidance on balancing a number of sample equations involving different elements and compounds.
What's hot (10)
Similar to 04 Solutions
Properties of Solution

1. The document discusses the properties of solutions including solutes, solvents, electrolytes, and non-electrolytes. Solutions are homogeneous mixtures of substances where one substance is in smaller amounts (solute) and the other is the continuous liquid medium (solvent).
2. The document also covers acid-base reactions and redox reactions that occur in aqueous solutions including precipitation, acid-base, and redox reactions. Precipitation reactions form insoluble products. Acid-base reactions involve proton transfer and redox reactions involve electron transfer.
3. Oxidation states and oxidation numbers are discussed as well as rules for determining oxidation numbers of elements in compounds. Redox reactions involve oxidation, where electrons
Chapter 8: Reactions in Aqueous Solution

This chapter discusses different types of chemical reactions in aqueous solutions. It introduces driving forces that cause reactions, such as formation of a solid, water, or gas. It explains how to predict products using solubility rules and oxidation-reduction reactions when metals react with nonmetals. Reactions are classified into double displacement, acid-base, single replacement, combustion, synthesis, or decomposition reactions based on their driving forces.
Ch4 Reactions in Aqueous Solution

This document discusses various topics related to aqueous solutions and reactions. It begins by defining key terms like solute, solvent, electrolyte and providing examples. It then covers properties of aqueous solutions such as conductivity. Various acid-base reactions and concepts are explained like Brønsted-Lowry acids and bases, neutralization reactions. Oxidation-reduction reactions and oxidation numbers are also discussed. Finally, the document covers concentration of solutions and calculations involving molarity, dilution and preparation of solutions.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
Periodic Properties Of Elements In The Periodic Table

The document summarizes periodic properties of elements in the periodic table, including periodic trends in atomic radius, ionization energy, electronegativity, and melting points. It also discusses periodic trends in chemical properties such as formulas of hydrides, oxides, and chlorides as well as their hydrolytic behaviors.
ACID_BASE.pptx

This document provides information on acid-base reactions and oxidation-reduction (redox) reactions. It defines acids and bases, and explains that in acid-base reactions, acids donate protons to bases. Neutralization reactions between acids and bases produce water and a salt. The document also discusses how to determine oxidation states of elements in compounds and identify the oxidized and reduced substances in redox reactions. It provides steps for balancing redox equations, including dividing the reaction into partial equations and adding electrons to balance charges. Examples of assigning oxidation states and balancing redox reactions are included.
ch4.ppt

1) A solution is a homogeneous mixture of two or more substances, where the solute is present in smaller amounts than the solvent.
2) An electrolyte is a substance that, when dissolved in water, allows the solution to conduct electricity through the formation of ions, while a nonelectrolyte does not form ions and the solution cannot conduct electricity.
3) Titrations involve slowly adding a solution of known concentration to another solution of unknown concentration until the chemical reaction between them is complete, as indicated by an indicator, and can be used to determine concentrations in acid-base and redox reactions.
Chapter 4 notes 

This document discusses aqueous solutions and their properties. It defines key terms including solute, solvent, solution, electrolyte, and nonelectrolyte. It explains that solutions can be solid, liquid, or gas and describes different types of aqueous solutions. Common examples like sea water, vinegar, and sugar water are provided. The document also discusses solubility, dissociation, hydration, and precipitation reactions.
10 Reduction And Oxidation

The document discusses oxidation-reduction (redox) reactions and oxidation numbers. It provides examples of half reactions and full redox reactions formed by combining half reactions. Oxidation involves an increase in oxidation state through loss of electrons, while reduction involves a decrease in oxidation state through gain of electrons. The species donating electrons is the reducing agent, while the species gaining electrons is the oxidizing agent.
Ch4 Reactions in Aqueous Solution (updated)

A document discusses various topics relating to chemistry solutions including:
1) The definition of solutions, solvents, and solutes. A solution is a homogeneous mixture of substances where the solute is the smaller component dissolved in the solvent.
2) Properties of aqueous solutions including that electrolytes can conduct electricity while nonelectrolytes cannot. Strong electrolytes dissociate completely while weak electrolytes only partially dissociate.
3) Reactions involving solutions such as precipitation reactions, acid-base reactions, and redox reactions. Precipitation occurs when an insoluble solid forms. Acid-base reactions involve acids and bases reacting to form water and a salt. Redox reactions involve the transfer of electrons
Classification of Reactions

The document classifies different types of chemical reactions:
1) Synthesis reactions involve combining two or more reactants to form one product.
2) Decomposition reactions involve breaking down one reactant into two or more products.
3) Single displacement reactions involve one compound exchanging ions with another.
4) Double displacement reactions involve two compounds exchanging ions to form two new compounds.
Similar to 04 Solutions (20)
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
pdfslide.net_redox-reactions-and-electrochemistry-redox-reactions-galvanic-ce...

pdfslide.net_redox-reactions-and-electrochemistry-redox-reactions-galvanic-ce...
Recently uploaded
Company Valuation webinar series - Tuesday, 4 June 2024

This session provided an update as to the latest valuation data in the UK and then delved into a discussion on the upcoming election and the impacts on valuation. We finished, as always with a Q&A
Income Tax exemption for Start up : Section 80 IAC

A presentation on the concept of Exemption of Profits of Start ups from Income Tax
How MJ Global Leads the Packaging Industry.pdf

MJ Global's success in staying ahead of the curve in the packaging industry is a testament to its dedication to innovation, sustainability, and customer-centricity. By embracing technological advancements, leading in eco-friendly solutions, collaborating with industry leaders, and adapting to evolving consumer preferences, MJ Global continues to set new standards in the packaging sector.
buy old yahoo accounts buy yahoo accounts

As a business owner, I understand the importance of having a strong online presence and leveraging various digital platforms to reach and engage with your target audience. One often overlooked yet highly valuable asset in this regard is the humble Yahoo account. While many may perceive Yahoo as a relic of the past, the truth is that these accounts still hold immense potential for businesses of all sizes.
Understanding User Needs and Satisfying Them

https://www.productmanagementtoday.com/frs/26903918/understanding-user-needs-and-satisfying-them
We know we want to create products which our customers find to be valuable. Whether we label it as customer-centric or product-led depends on how long we've been doing product management. There are three challenges we face when doing this. The obvious challenge is figuring out what our users need; the non-obvious challenges are in creating a shared understanding of those needs and in sensing if what we're doing is meeting those needs.
In this webinar, we won't focus on the research methods for discovering user-needs. We will focus on synthesis of the needs we discover, communication and alignment tools, and how we operationalize addressing those needs.
Industry expert Scott Sehlhorst will:
• Introduce a taxonomy for user goals with real world examples
• Present the Onion Diagram, a tool for contextualizing task-level goals
• Illustrate how customer journey maps capture activity-level and task-level goals
• Demonstrate the best approach to selection and prioritization of user-goals to address
• Highlight the crucial benchmarks, observable changes, in ensuring fulfillment of customer needs
Hamster Kombat' Telegram Game Surpasses 100 Million Players—Token Release Sch...

Hamster Kombat' Telegram Game Surpasses 100 Million Players—Token Release Schedule Unveiled
ModelingMarketingStrategiesMKS.CollumbiaUniversitypdf

Implicitly or explicitly all competing businesses employ a strategy to select a mix
of marketing resources. Formulating such competitive strategies fundamentally
involves recognizing relationships between elements of the marketing mix (e.g.,
price and product quality), as well as assessing competitive and market conditions
(i.e., industry structure in the language of economics).
Best Forex Brokers Comparison in INDIA 2024

Navigating the world of forex trading can be challenging, especially for beginners. To help you make an informed decision, we have comprehensively compared the best forex brokers in India for 2024. This article, reviewed by Top Forex Brokers Review, will cover featured award winners, the best forex brokers, featured offers, the best copy trading platforms, the best forex brokers for beginners, the best MetaTrader brokers, and recently updated reviews. We will focus on FP Markets, Black Bull, EightCap, IC Markets, and Octa.
Top mailing list providers in the USA.pptx

Discover the top mailing list providers in the USA, offering targeted lists, segmentation, and analytics to optimize your marketing campaigns and drive engagement.
Best practices for project execution and delivery

A select set of project management best practices to keep your project on-track, on-cost and aligned to scope. Many firms have don't have the necessary skills, diligence, methods and oversight of their projects; this leads to slippage, higher costs and longer timeframes. Often firms have a history of projects that simply failed to move the needle. These best practices will help your firm avoid these pitfalls but they require fortitude to apply.
Organizational Change Leadership Agile Tour Geneve 2024

Organizational Change Leadership at Agile Tour Geneve 2024
Structural Design Process: Step-by-Step Guide for Buildings

The structural design process is explained: Follow our step-by-step guide to understand building design intricacies and ensure structural integrity. Learn how to build wonderful buildings with the help of our detailed information. Learn how to create structures with durability and reliability and also gain insights on ways of managing structures.
2024-6-01-IMPACTSilver-Corp-Presentation.pdf

IMPACT Silver is a pure silver zinc producer with over $260 million in revenue since 2008 and a large 100% owned 210km Mexico land package - 2024 catalysts includes new 14% grade zinc Plomosas mine and 20,000m of fully funded exploration drilling.
Creative Web Design Company in Singapore

At Techbox Square, in Singapore, we're not just creative web designers and developers, we're the driving force behind your brand identity. Contact us today.
Call 8867766396 Satta Matka Dpboss Matka Guessing Satta batta Matka 420 Satta...

CALL 8867766396 SATTA MATKA | DPBOSS | KALYAN MAIN BAZAR | FAST MATKA | DPBOSS GUESSING | TARA MATKA | KALYAN CHART | MATKA BOSS
Recently uploaded (20)
Company Valuation webinar series - Tuesday, 4 June 2024

Company Valuation webinar series - Tuesday, 4 June 2024
Income Tax exemption for Start up : Section 80 IAC

Income Tax exemption for Start up : Section 80 IAC
Hamster Kombat' Telegram Game Surpasses 100 Million Players—Token Release Sch...

Hamster Kombat' Telegram Game Surpasses 100 Million Players—Token Release Sch...
ModelingMarketingStrategiesMKS.CollumbiaUniversitypdf

ModelingMarketingStrategiesMKS.CollumbiaUniversitypdf
Organizational Change Leadership Agile Tour Geneve 2024

Organizational Change Leadership Agile Tour Geneve 2024
Structural Design Process: Step-by-Step Guide for Buildings

Structural Design Process: Step-by-Step Guide for Buildings
Call 8867766396 Satta Matka Dpboss Matka Guessing Satta batta Matka 420 Satta...

Call 8867766396 Satta Matka Dpboss Matka Guessing Satta batta Matka 420 Satta...
The Influence of Marketing Strategy and Market Competition on Business Perfor...

The Influence of Marketing Strategy and Market Competition on Business Perfor...
04 Solutions
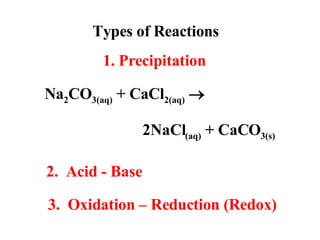
- 1. Types of Reactions Na 2 CO 3(aq) + CaCl 2(aq) 2NaCl (aq) + CaCO 3(s) 1. Precipitation 2. Acid - Base 3. Oxidation – Reduction (Redox)
- 2. Acids & Bases (Bronsted) Acid = Proton (H + ) donor Base = Proton acceptor Acid Properties: sour (vinegar and citrus fruit) turns litmus red
- 3. Strong Acids HCl, HNO 3 , H 2 SO 4 HCl (aq) H + (aq) + Cl - (aq) or ….. HCl + H 2 O (l) H 3 O + (aq) + Cl - (aq) There is no HCl present in solution proton hydronium ion monoprotic diprotic
- 4. Weak Acids HCN (aq) H + (aq) + CN - (aq) HCN, H + and CN - are present in solution H 2 O (l) H + (aq) + OH - (aq) All aqueous solutions will contain protons and hydroxide ions. Acetic acid = H(C 2 H 3 O 2 ) Phosphoric Acid = H 3 PO 4
- 5. Base Properties: bitter slippery turns litmus blue Strong bases: NaOH; KOH Weak bases: NH 3 , CO 3 2-
- 6. Neutralization Reactions Acid + Base salt + H 2 O HNO 3(aq) + KOH (aq) KNO 3(aq) + H 2 O (l) HCl (aq) + Na 2 CO 3(aq) KNO 3(aq) + CO 2(g) + H 2 O (l)
- 7. Oxidation Reduction Reactions (Redox) Transfer of electrons (between atoms in compounds) oxidation = loss of electrons reduction = gain of electrons oxidizing agent – gets reduced by accepting electrons reducing agent – gets oxidized by donating electrons
- 8. Oxidation State - Rules 1. 0 in elemental form (e.g. H 2 , O 2 , Zn (s) ) 2. Ion charge = oxidation state for ions (of elements ) 3. O = -2 (except in peroxides, e.g. H 2 O 2 ) 4. H = +1 (except as hydride in ionic cpd) 5. F = -1 (other halides = -1 if ionic cpd) 6. Sum of ox #s = charge of ion or 0 for neutral cpd
- 9. Oxidation-Reduction Reactions Any combustion reaction cpd of (CHO) + O 2 CO 2 + H 2 O example methane combustion …. Reaction of acid with metals …. 2HCl (aq) + Zn (s) ZnCl 2(aq) + H 2(g) __Al (s) + __Fe 2 O 3(s) __Fe (s) + __Al 2 O 3(s) + heat
- 11. Blood Alcohol Levels Legal limit = 0.1% by mass/(weight) What is approximate Molarity? ethanol ~ 46.0 g mol -1 How much dilution of 3.2% beer results in this? Why is this misleading?
- 12. Breath Analyzer ___C 2 H 6 O + ___ K 2 Cr 2 O 7 + ___ H 2 SO 4 ___C 2 H 4 O 2 + ___ Cr 2 (SO 4 ) 3 + ___ K 2 SO 4 + ___ H 2 O
- 13. Getting Mg from seawater 1. CaCO 3(s) CaO (s) + CO 2(g) 2. CaO (s) + H 2 O (l) + Ca 2+ (aq) + 2OH - (aq) 3. Mg 2+ (aq) + 2OH - (aq) MgOH 2(s) 4. MgOH 2(s) + HCl (aq) MgCl 2(aq) + 2H 2 O (l) 5. MgCl 2(l) + e - Mg (l) + Cl 2(g)