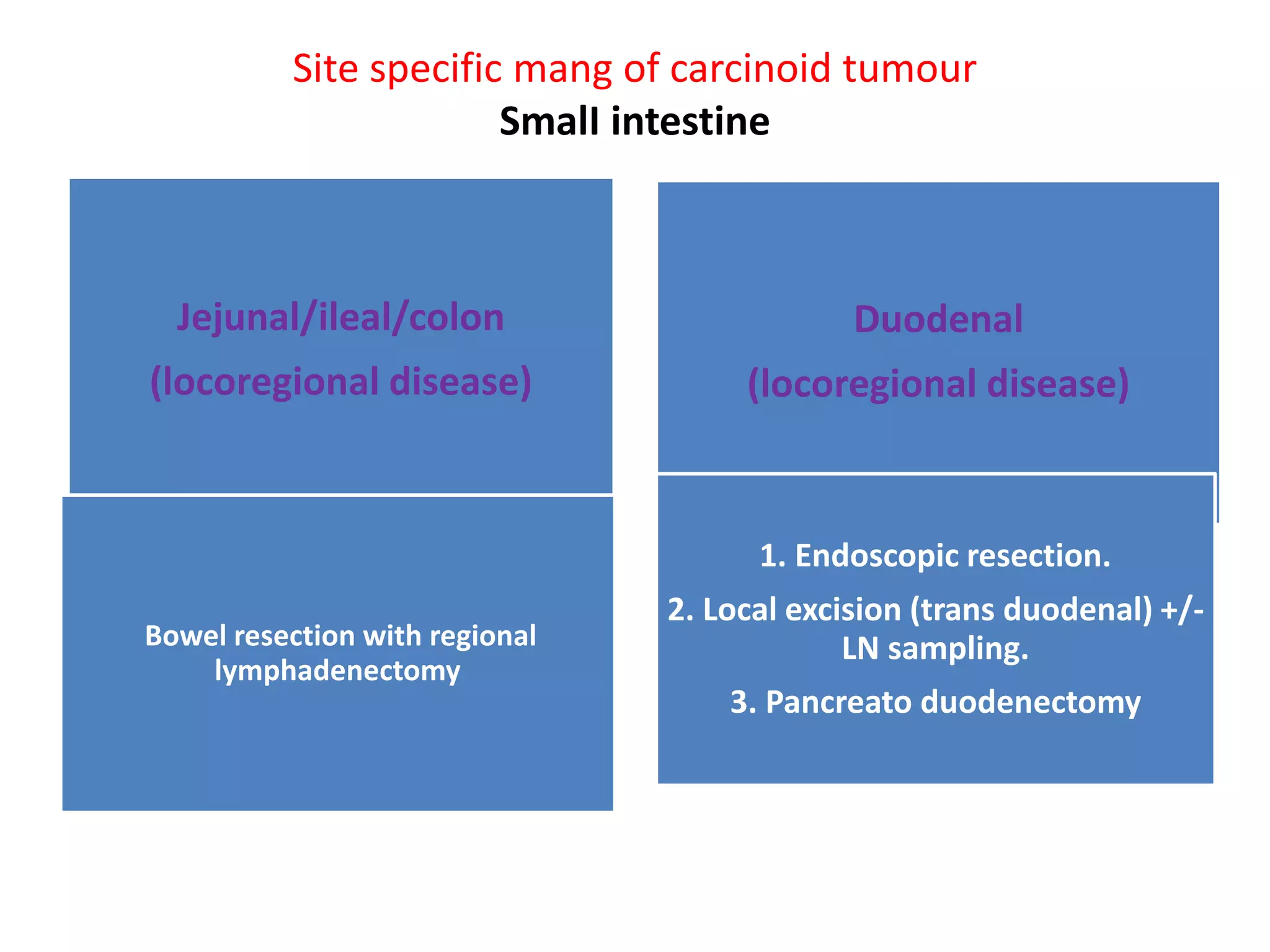
Carcinoid tumors are slow-growing neuroendocrine tumors that commonly arise in the gastrointestinal tract and lungs. The document discusses carcinoid tumors in depth, including their definition, sites of origin, histology, staging, clinical features, diagnostic testing, and management approaches. Treatment involves surgical resection when possible, with additional therapies for advanced or metastatic disease aimed at controlling hormone secretion and tumor growth.





























































![• outward growth of many GISTs within the
gastrointestinal wall is one of the reasons why
several are diagnosed relatively late,
• One-fourth of GISTs are discovered
incidentally during diagnostic assessments
(whether an endoscopic procedure,
ultrasound, or computed tomography [CT]
scan) done for other reasons.](https://image.slidesharecdn.com/carcinoid-171212163358/75/Carcinoid-tumors-63-2048.jpg)







































