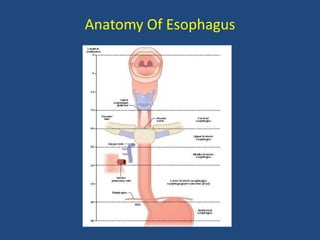
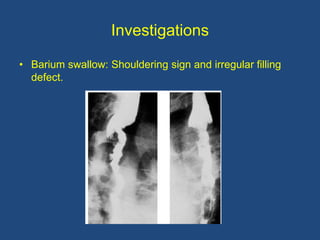
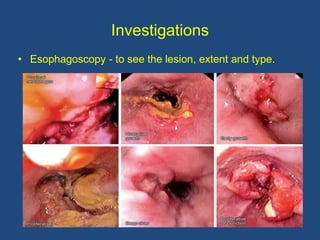
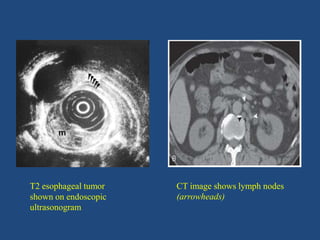
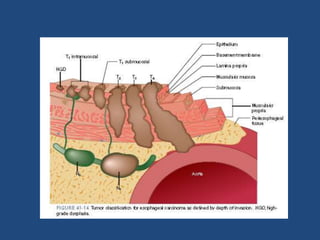
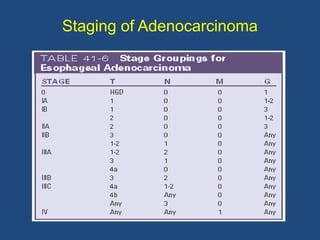
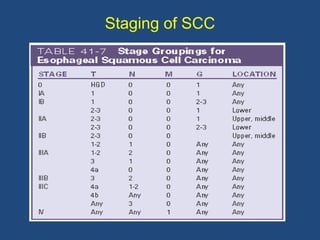
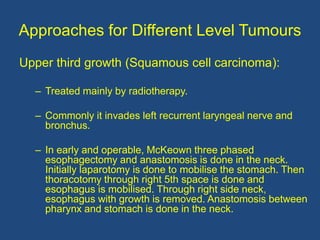
This document provides information about carcinoma of the esophagus, including its epidemiology, risk factors, pathological classification, clinical features, investigations, diagnosis and staging, and treatment. Carcinoma of the esophagus is most common in China, South Africa, and parts of India. It typically presents with dysphagia. Investigations include endoscopy with biopsy, imaging like CT and PET scans, and endoscopic ultrasound. Treatment depends on the stage, with surgery or chemoradiation used for early-stage or locally advanced carcinoma, and palliative approaches for metastatic disease.