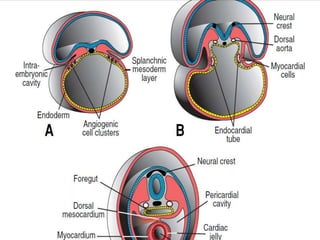
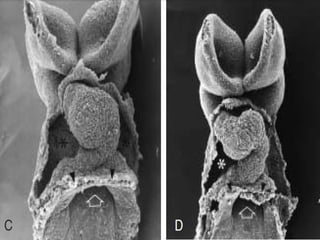
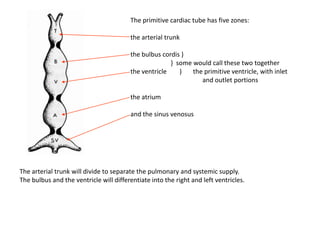
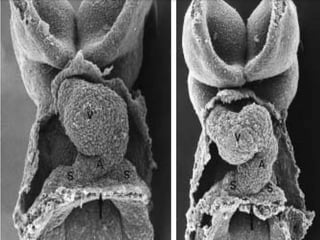
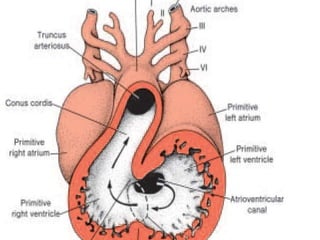
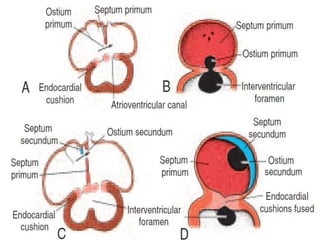
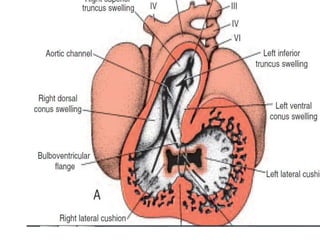
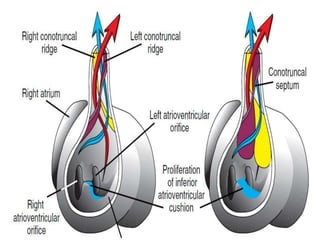
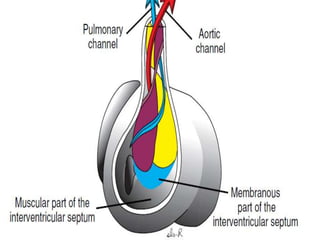
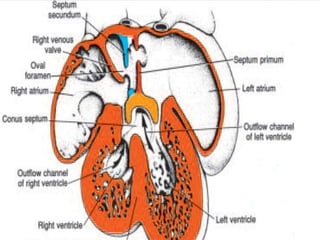
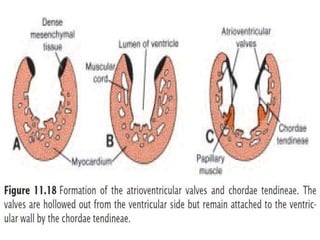
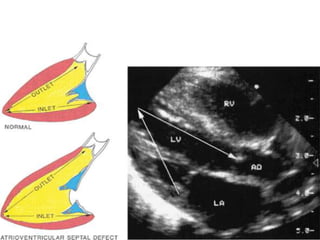
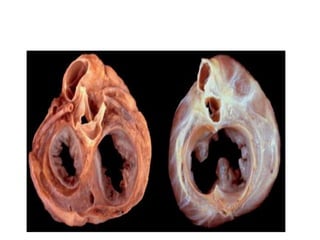
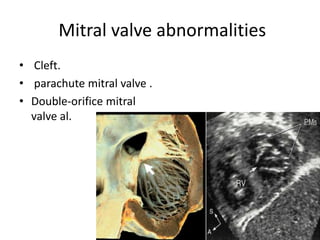
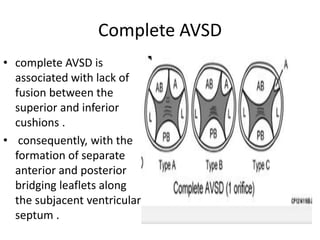
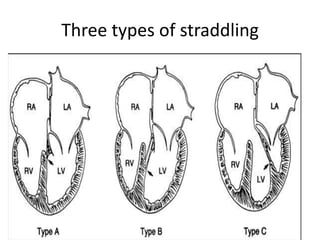
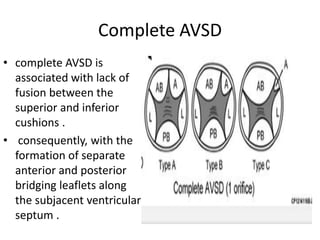
This document discusses the embryology, anatomy, classifications, and associated anomalies of atrioventricular septal defects (AVSDs). It describes the embryologic development of endocardial cushions and septum that can lead to partial or complete AVSD. Partial AVSD involves a cleft in the anterior mitral valve leaflet while complete AVSD lacks fusion of endocardial cushions, forming a common atrioventricular valve. Complete AVSD is further classified and associated anomalies are discussed, including conotruncal defects, coronary anomalies, and ventricular disproportion. Valve abnormalities like double orifice, papillary muscle anomalies, and their implications are also summarized.






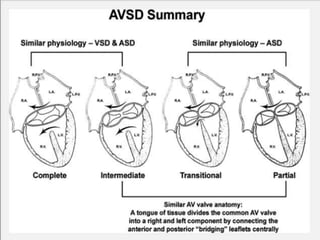
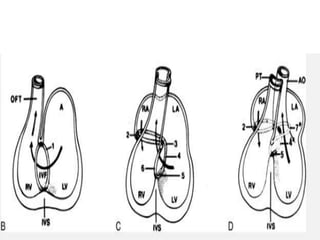
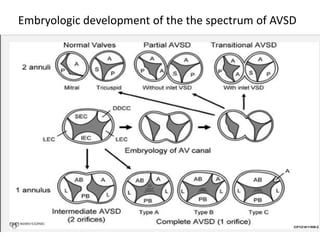


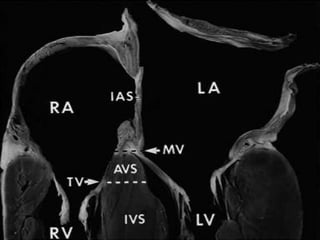
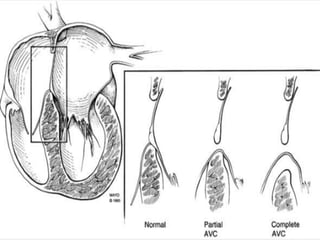







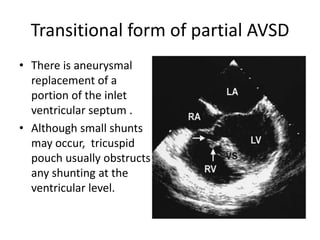

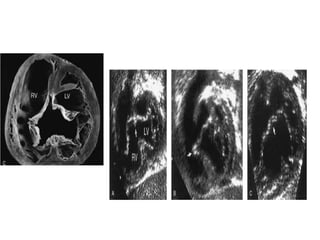




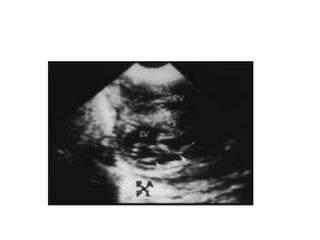



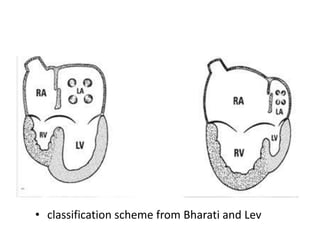
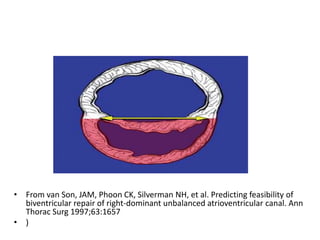




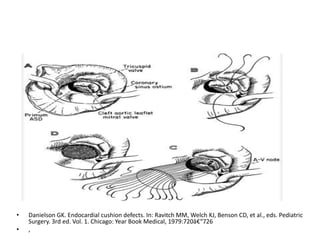


















![Reoperation after Repair of partial Atrioventricular Septal Defects
Regurgitation or stenosis of the left
atrioventricular valve [ 10% to 15%].
Subaortic stenosis.
Residual recurrent ASD.](https://image.slidesharecdn.com/avsd-130120231854-phpapp02/85/atio-ventricular-septal-defects-75-320.jpg)



































