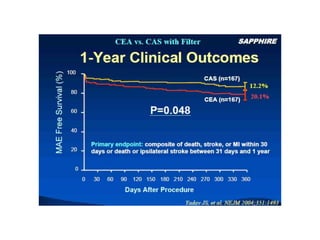
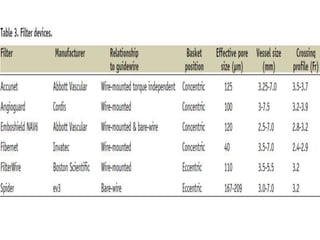
Carotid artery disease is a major cause of stroke. Left untreated, carotid stenosis over 75% carries a risk of stroke of 2-5% per year. Carotid endarterectomy has been shown in clinical trials such as NASCET and ACAS to significantly reduce stroke risk compared to medical management alone, with perioperative stroke or death rates of less than 6% for symptomatic patients and 3% for asymptomatic patients. Carotid artery stenting is an alternative treatment that utilizes embolic protection devices and stent placement to treat carotid stenosis, but requires technical expertise to achieve outcomes comparable to surgery.