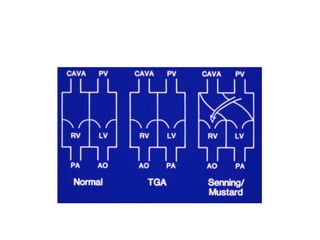
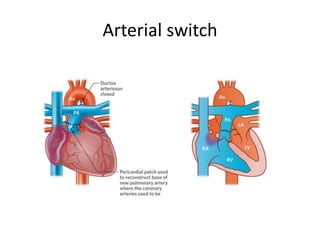
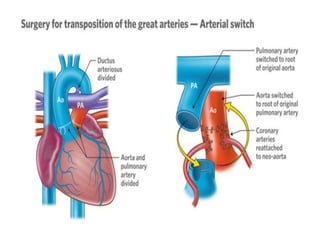
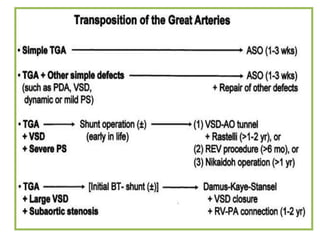
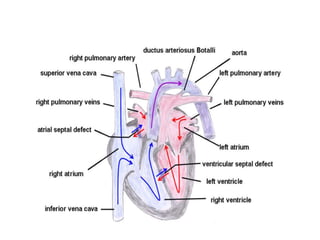
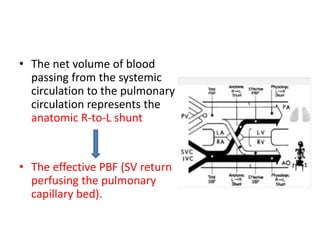
D-Transposition, also known as dextro-Transposition of the great arteries (d-TGA), is a congenital heart defect where the ventricles are connected to the wrong great arteries. Specifically, the aorta arises from the right ventricle while the pulmonary artery arises from the left ventricle. This causes two parallel circulations instead of the normal series circulation. The basic embryological defect is abnormal development of the conus, which prevents normal septal formation between the great arteries. Untreated d-TGA is fatal in infancy due to lack of oxygenated blood to the body. Clinical presentation depends on the degree of mixing between the circulations.






























![Coronary Arteries
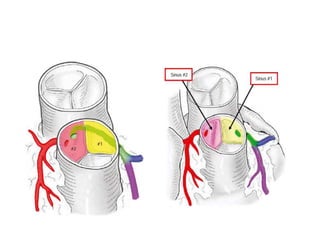
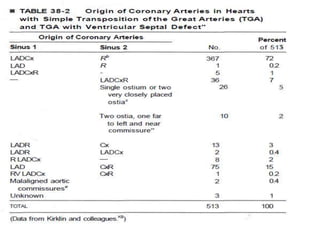
• aortic sinuses that face the pulmonary trunk,
regardless of the interrelationships of the great
arteries
• LAD and LCx arise as a single trunk (LCA]) from aortic
sinus 1 and distribute in a normal manner
• RCA = sinus 2 and follows this artery's usual course.
• single coronary artery= sinus 2.](https://image.slidesharecdn.com/dtga-180722050527/85/D-TGA-34-320.jpg)





























![]TGA and essentially intact ventricular septum
80% at 1 week
only 17% at 2 months
4% at 1 year
better when there is a true ASD.
TGA and important VSD
• 91% at 1 month
• 43% at 5 months
• 32% at 1 year
• lower when the patient has a very large Qp.](https://image.slidesharecdn.com/dtga-180722050527/85/D-TGA-66-320.jpg)