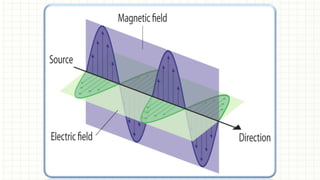
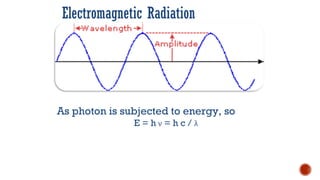
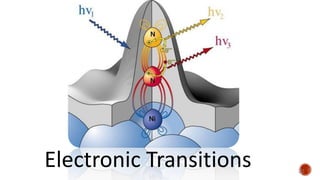
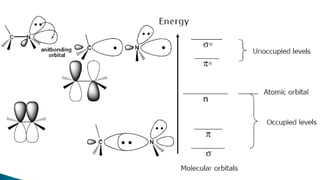
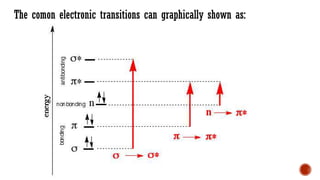
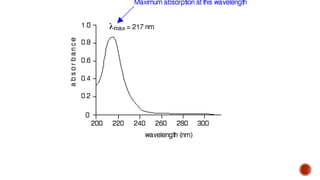
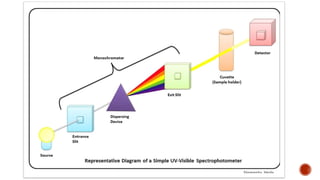
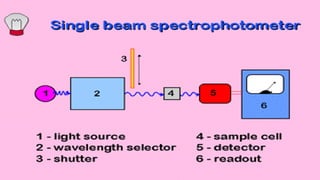
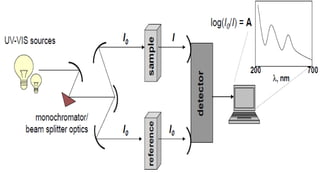
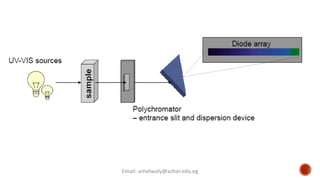
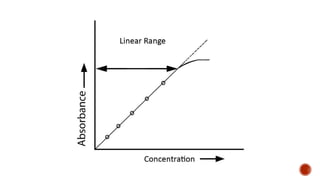
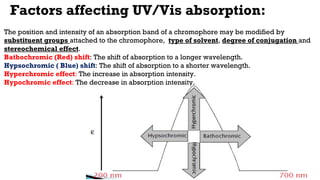
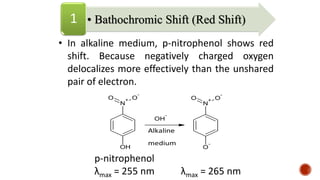
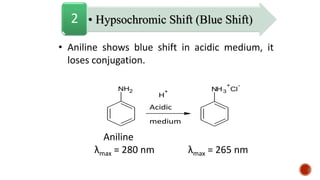
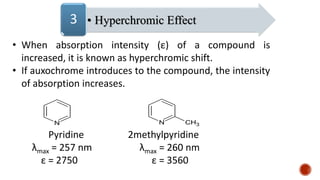
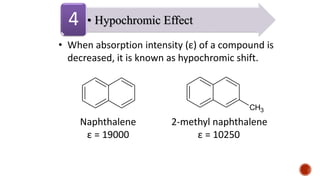
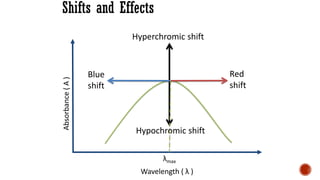
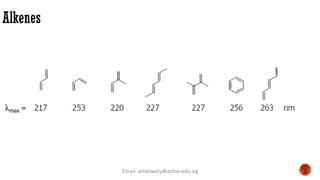
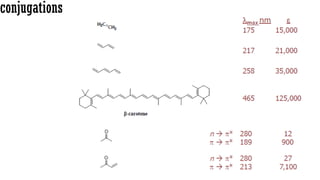
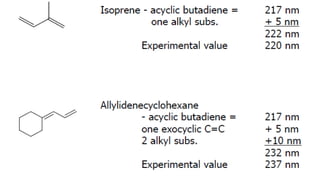
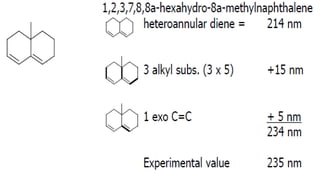
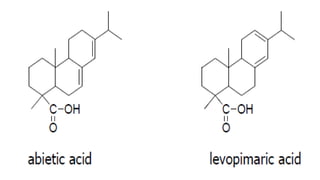
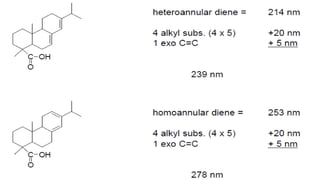
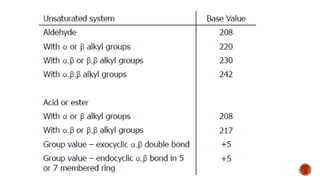
The document presents an overview of ultraviolet/visible (UV/Vis) spectroscopy, including the principles, instrumentation, and applications of this technique. It describes the electromagnetic radiation involved, the significance of photon energy absorption, and the factors affecting absorption, such as chromophores and auxochromes. Additionally, it outlines various shifts (bathochromic, hypsochromic, hyperchromic, and hypochromic) and their implications in spectroscopic analysis.