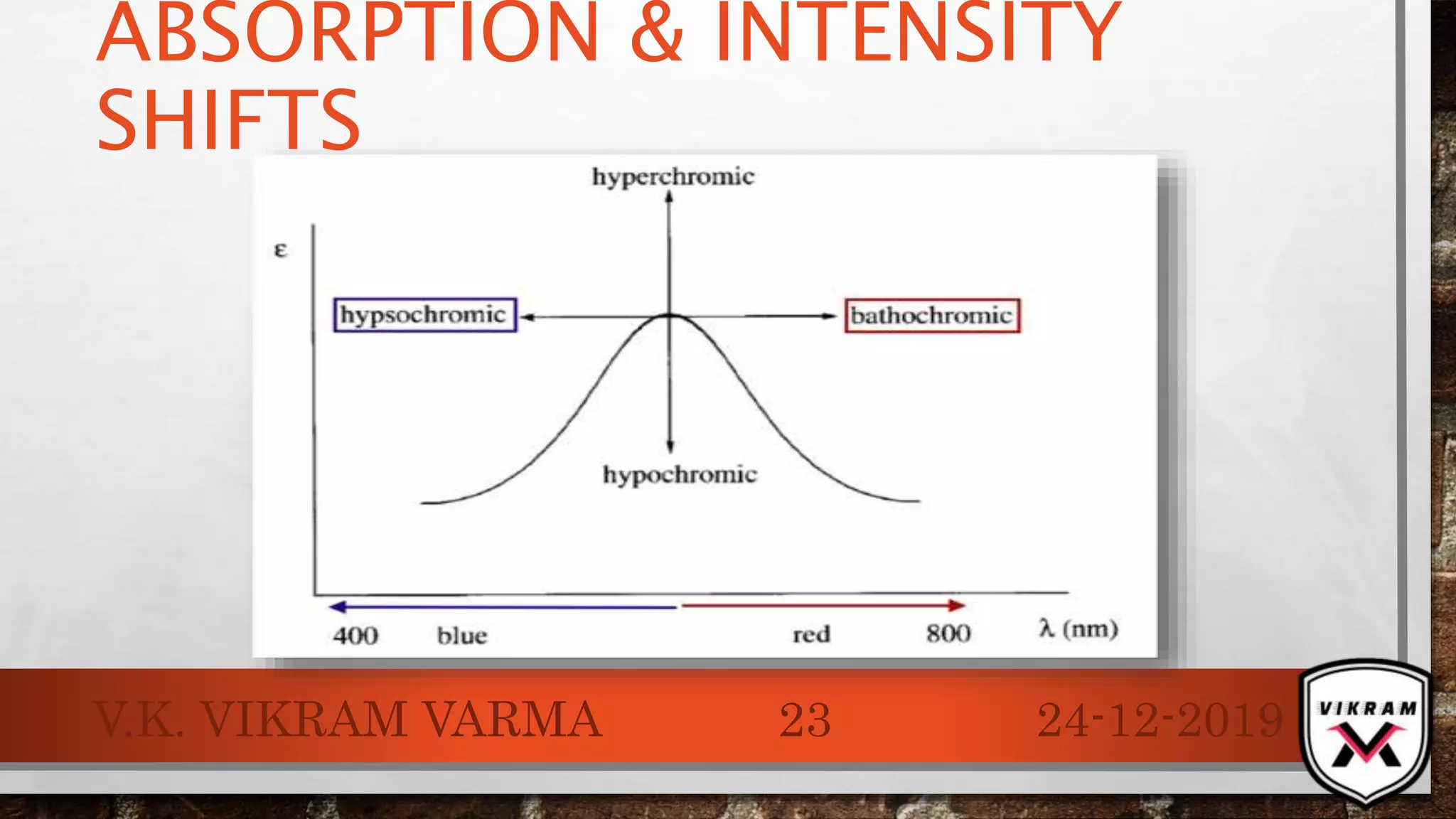
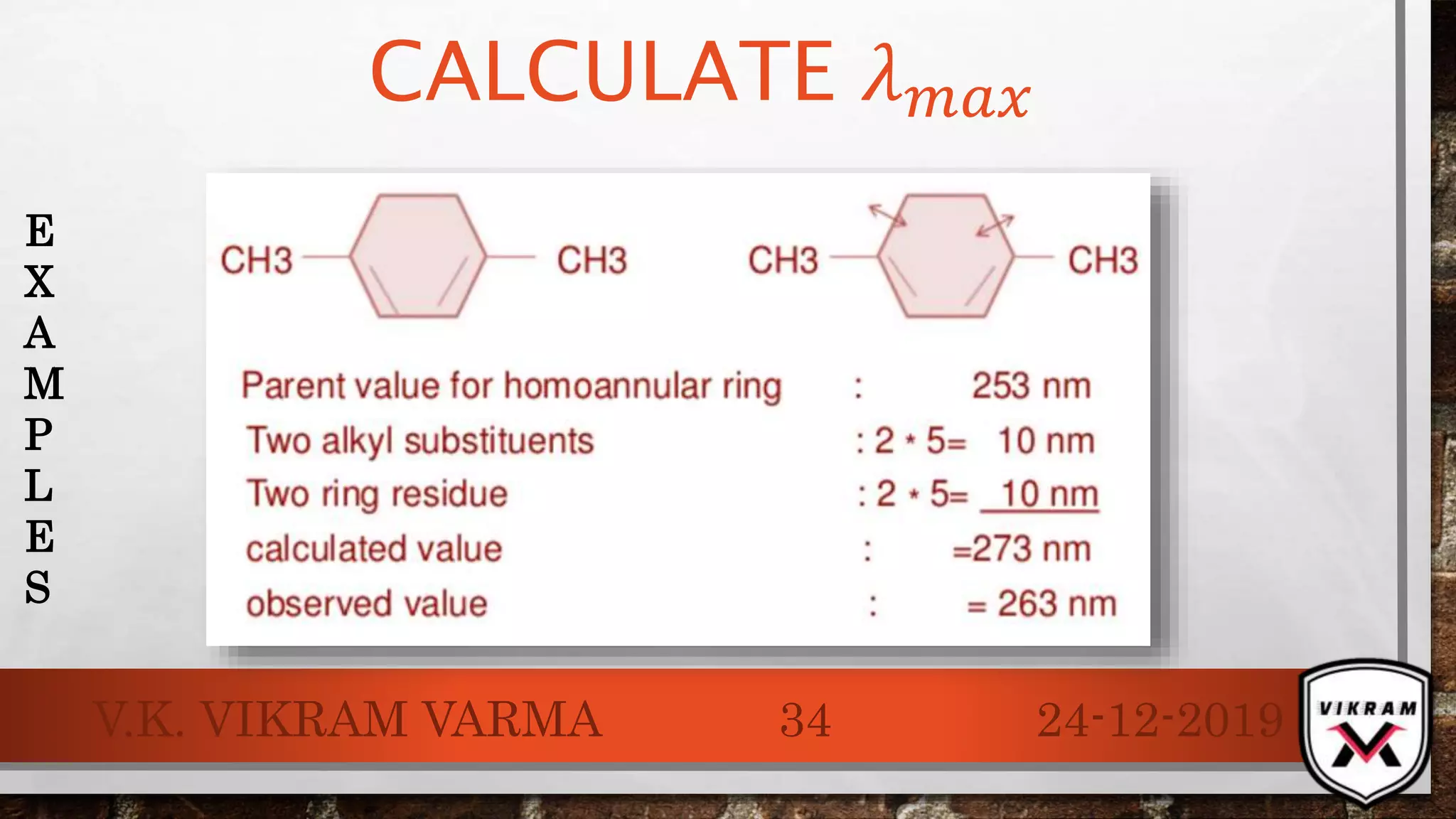
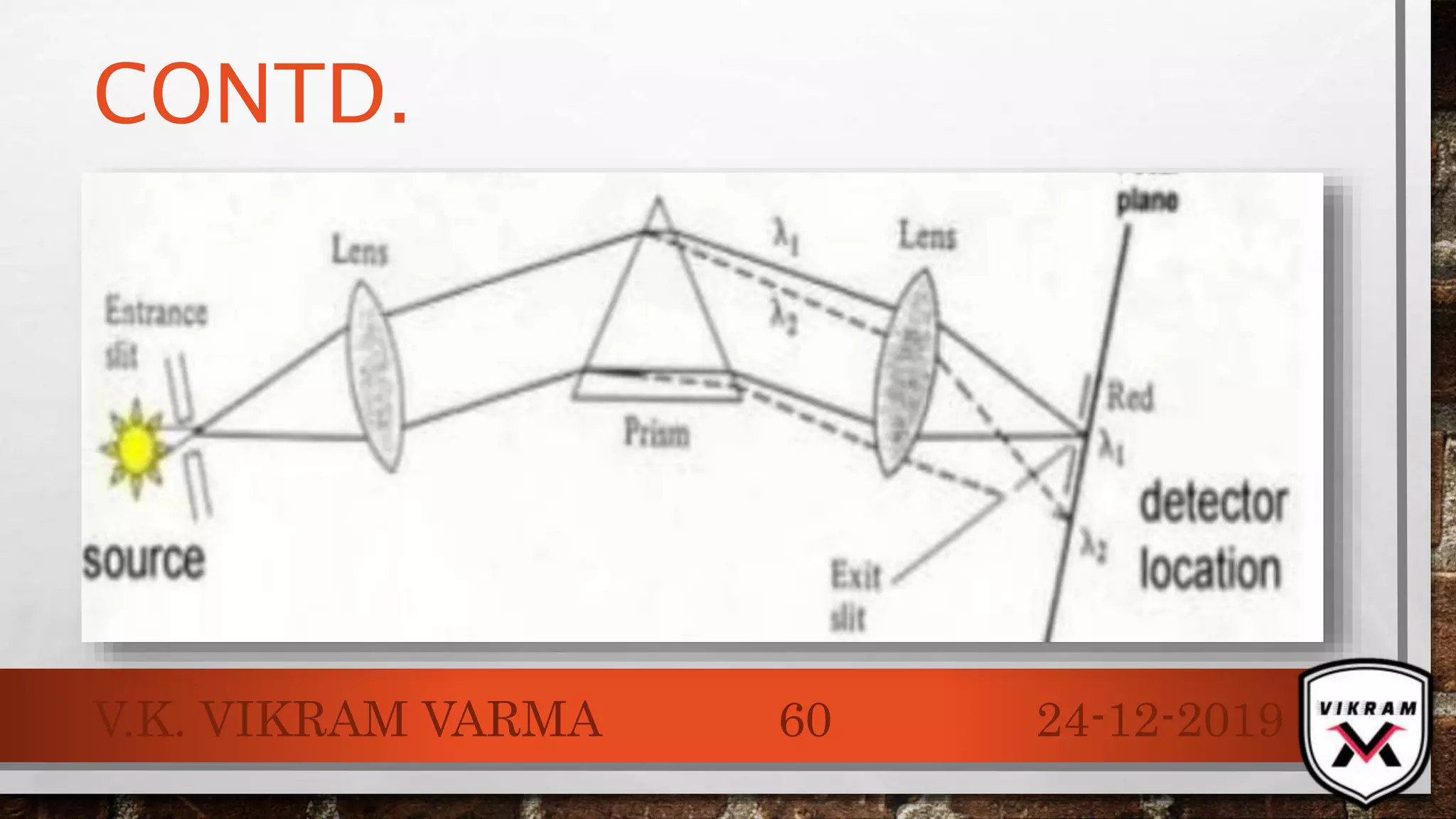
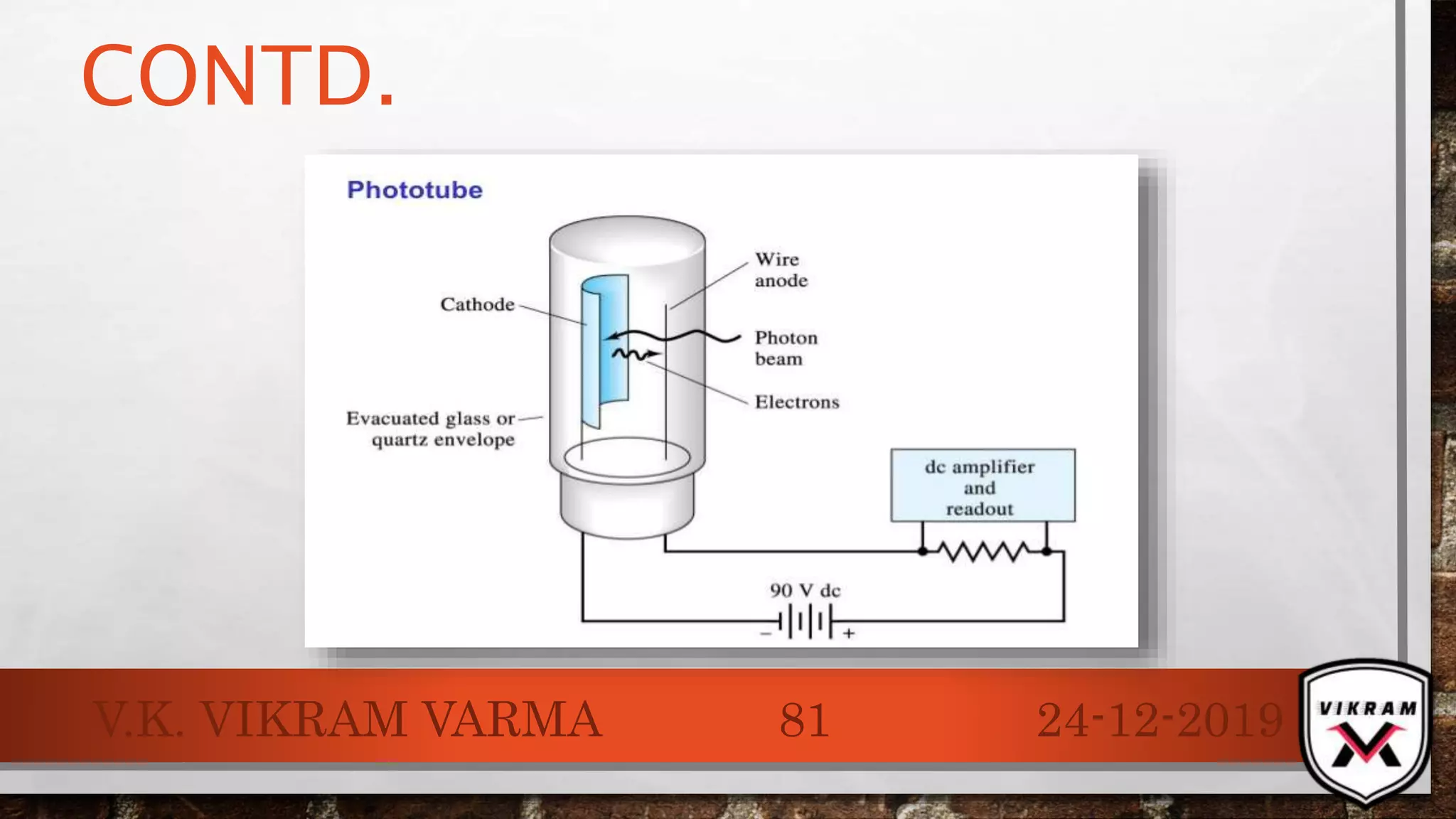
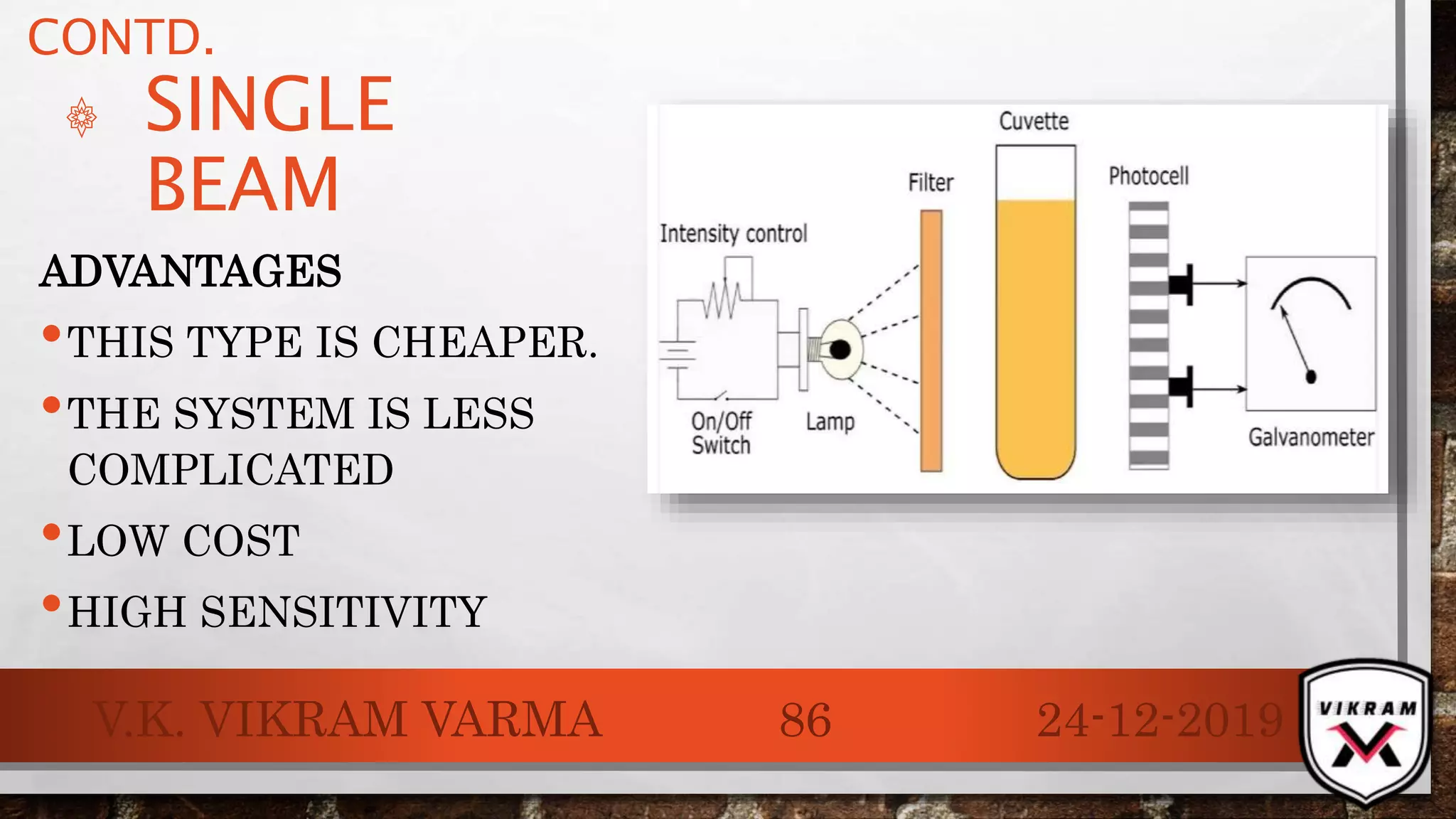
The document discusses UV-visible spectroscopy, outlining its principles, instrumentation, and applications. It explains the interactions of electromagnetic radiation with matter, laws governing absorption, and the Woodward-Fieser rule for calculating maximum absorption wavelengths based on molecular structure. Various details regarding the instrumentation components, such as light sources and wavelength selectors, are also included.