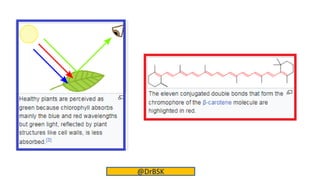
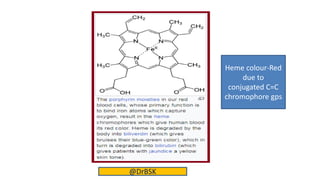
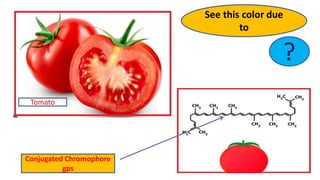
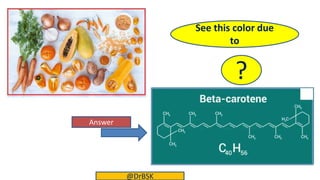
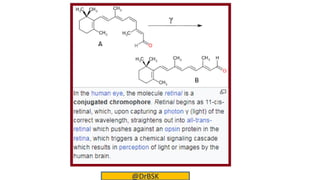
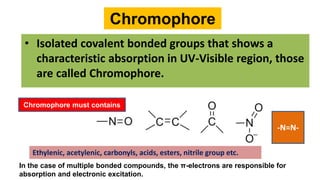
The document explains the concepts of chromophores and auxochromes, highlighting that chromophores are covalently bonded groups with characteristic absorption in the UV-visible region, while auxochromes do not absorb light but can enhance absorption when attached to chromophores. It provides examples of both, detailing how auxochromes can shift absorption wavelengths and intensify colors in organic compounds. The text also illustrates these concepts with specific examples such as nitrobenzene and aniline, showing how their respective functional groups influence color.