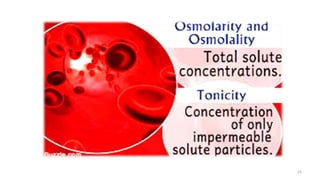
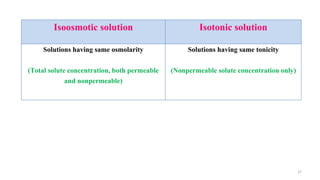
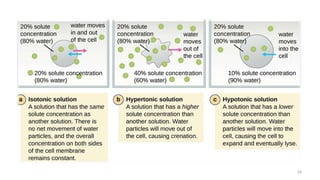
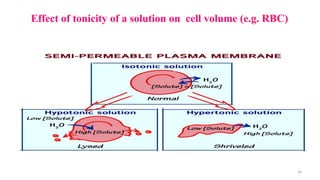
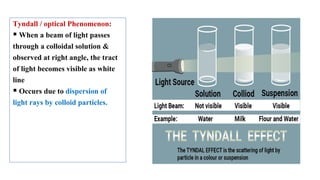
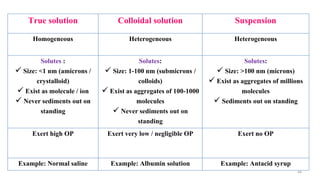
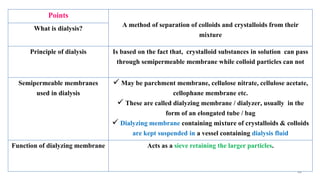
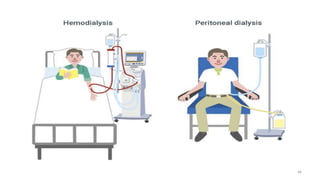
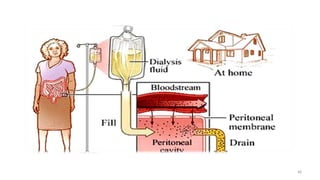
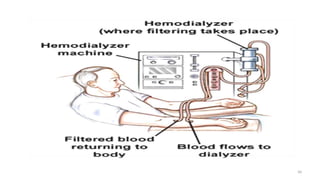
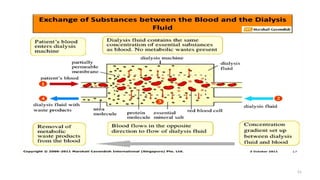
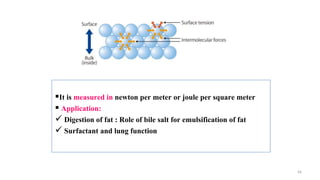
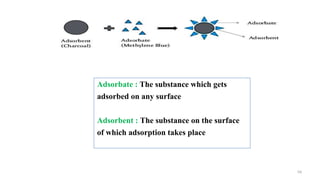
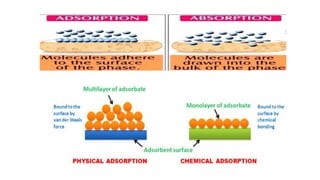
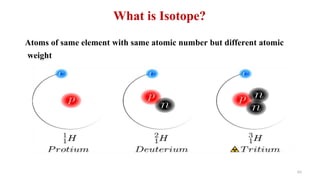
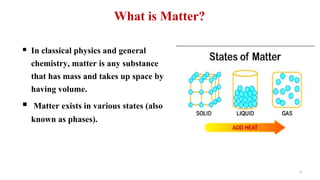
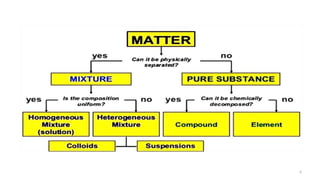
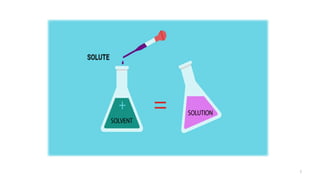
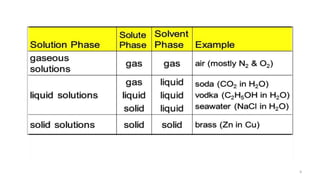
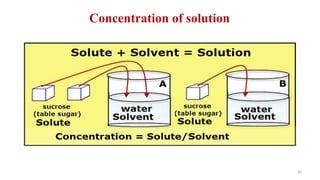
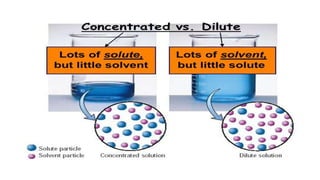
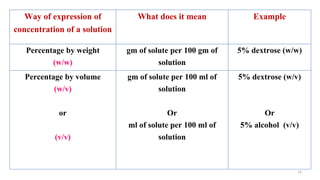
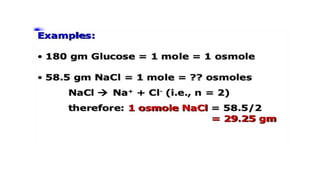The document provides an extensive overview of solutions, their properties, and classifications, focusing on concepts such as solutes, solvents, and types of mixtures. It explains various ways to express concentration, the significance of osmolarity and tonicity, as well as the distinctions between crystalloid and colloidal solutions. Additionally, it covers topics like dialysis, osmosis, surface tension, viscosity, and isotopes.





![15
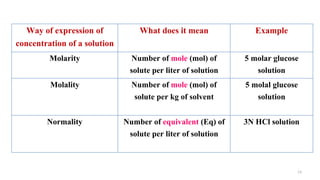
Mole: (Mol or mol)
Molecular weight or atomic weight
of a substance expressed in gram
gm-MW (if compound)
or gm-AW (if element)
Equivalent: (Eq or eq)
The equivalent weight of a substance
expressed in gram
Equivalent weight = MW or AW / valency
[ If acid, Eq = MW / Number of “H” atoms]
So, Eq = Mole / Valency
Osmole: (Osm or osm)
Amount of osmotically active substances in gram, which in 1 liter solution with water,
exerts an OP of 22.4 atmosphere (22.4 x 760 mm Hg) & depresses the freezing point of
water by 1.86 degree C.
Osmole = Mole / Number of particle given by each molecule in solution by dissociation](https://image.slidesharecdn.com/solutioncrystalloidscolloidsisotopeformphilpaperi-200928054459/85/Solution-crystalloids-colloids-isotope-15-320.jpg)