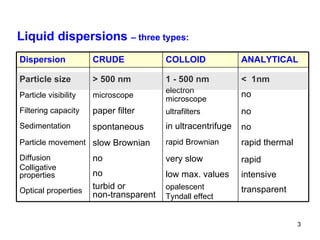
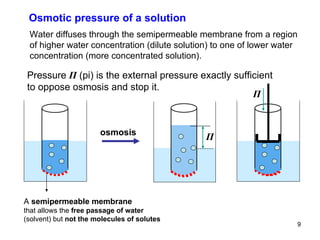
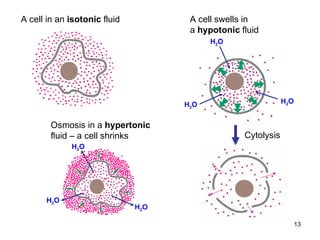
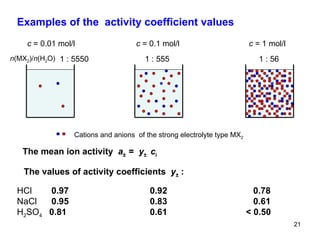
This document provides an overview of solutions and their properties. It discusses the components and types of liquid dispersions including analytical, colloid, and crude dispersions. It also describes concentration measures like molarity and molality. Key properties discussed include osmotic pressure, boiling point elevation, freezing point depression, and vapor pressure lowering. The document examines electrolytes, nonelectrolytes, and weak/strong electrolytes. It also discusses isotonic, hypertonic, and hypotonic solutions as well as their effects on cell volume.










![Blood plasma osmolality is measured by means of osmometers. Blood plasma osmolality 280 – 295 mmol kg H 2 O –1 Hypoosmolality (up to 230 mmol/kg) – deficit in Na + or hyperhydration Hyperosmolality (up to 400 mmol/kg) – retention of Na + , dehydration, hyperglycaemia, uremic syndrome, unusual compounds (e.g. ethanol, ethylene glycol, acetone). It is under the strict hormonal control (aldosterone, vasopressin, atrial natriuretic peptides). Plasma osmolality (mmol kg H 2 O -1 ) ≈ 2 [ Na + ] + [ glucose ] + [ urea] (mmol/l) or ≈ 1.86 [Na + ] + [glucose] + [urea] + 9 (mmol/l) The marked difference between the measured and the roughly estimated value is the sign of the " osmolar gap“ that is usually caused by high concentration of other unionized compou n d s (ethanol, acetone, etc.). In spite of the known value of osmolality gained by measurement, it is useful to calculate the rough estimate of plasma osmolality from the values of major plasma solutes:](https://image.slidesharecdn.com/01solutionselectrolytesprotolyticequilibria-110914014716-phpapp02/85/01-solutions-_electrolytes__protolytic_equilibria-14-320.jpg)













![Ionic product of water K w From the quantitative point of view, the ionization of water is described by the equilibrium ionization constant K c : Because the high value of unionized molecules of water [H 2 O] (55.5 mol/l) changes only slightly in real solutions, instead of K c the constant ionic product of water K w is used: In any aqueous solutions, concentrations [ H + ] and [ OH – ] can achieve only those values that are in agreement with the K w value 1.00 10 –14 . K c [H + ] [OH – ] [H 2 O] K c [H 2 O] = K w = [ H + ] [ OH – ] = 1.00 10 –14 (25 °C)](https://image.slidesharecdn.com/01solutionselectrolytesprotolyticequilibria-110914014716-phpapp02/85/01-solutions-_electrolytes__protolytic_equilibria-30-320.jpg)
![In pure water , the concentrations [H + ] and [OH – ] are the same and the concentration of each of this ions is equal to 1.00 10 –7 mol/l (at 25 °C) . All aqueous solution in which [H + ] equals [OH–] are neutral . In acidic solutions [H+] is greater than [OH – ], in alkaline solutions (basic solutions) [OH – ] > [H + ]. Based upon Le Chatelier´s principle, adding H + to a neutral aqueous solution will decrease [OH – ] and similarly, adding OH – will decrease [H + ]. A more convenient way for expressing [H + ] and [OH – ] in mol/l (small numbers, scientific notation) are the expressions pH and pOH defined as the negative logarithms of [H + ] and [OH – ]: pH = – log [ H + ] and pOH = – log [ OH – ]](https://image.slidesharecdn.com/01solutionselectrolytesprotolyticequilibria-110914014716-phpapp02/85/01-solutions-_electrolytes__protolytic_equilibria-31-320.jpg)
![The values of pH and pOH for dilute aqueous solutions fall between 0 and 14 . The same logarithmic notation can be also used for K w : K w = [H + ] [OH – ] = 1.00 10 –14 p K w = pH + pOH = 14 so that pH = 14 – pOH The pH and pOH scales are logarithmic, not linear, scales ! A change of pH by 1 represents the 10 times higher or lower concentration of [H + ]; any two-fold increase in [H+] results in the decrease of pH by 0.3, because the logarithm of 2 equals 0.30.](https://image.slidesharecdn.com/01solutionselectrolytesprotolyticequilibria-110914014716-phpapp02/85/01-solutions-_electrolytes__protolytic_equilibria-32-320.jpg)
![Dissociation of a strong acid: HA + H 2 O H 3 O + + A – In solutions of monoprotic strong acids , the concentration [H + ] is equal to the total strong acid concentration c HA : [ H + ] = c HA and pH = – log [ H + ] = – log c HA Dissociation of a strong hydroxide: MeOH( aq ) Me + + OH – In solutions of monobasic strong hydroxides , [ OH – ] = c MeOH , pOH = – log [OH – ] = – log c MeOH , and pH = 14 – pOH pH of strong acids and strong hydroxides solutions Strong acid and strong hydroxides are strong electrolytes that are fully dissociated in aqueous solutions. (In diluted solutions, the difference between the concentration and the activity of ions can be neglected.) Let us remind the list of a few strong acids and hydroxides!](https://image.slidesharecdn.com/01solutionselectrolytesprotolyticequilibria-110914014716-phpapp02/85/01-solutions-_electrolytes__protolytic_equilibria-33-320.jpg)
![The titration curve of a monoprotic strong acid ( c HA = 0.1 mol/l) Linear decrease in [H + ] in the course of titration results in a logarithmic curve representing the increase of pH values. To reach the pH value greater by 1 than the initial, 90 % of the amount of an strong acid have to be neutralized. 12 10 8 6 4 2 0 pH pH 7.00 (excess NaOH) n ( OH – ) / n ( acid ) 0 0.2 0.4 0.6 0.8 1.0](https://image.slidesharecdn.com/01solutionselectrolytesprotolyticequilibria-110914014716-phpapp02/85/01-solutions-_electrolytes__protolytic_equilibria-34-320.jpg)
![Dissociation of weak electrolytes, ( of weak acids and weak bases ) A weak monoprotic acid A weak base Equilibrium constants of ionization Acid and base ionization constants Weak electrolytes are ionized to only a slight extent, the concentration of ions are relatively low; most solute molecules do not split into ions. HA + H 2 O H 3 O + + A – B + H 2 O BH + + OH – K c [H 3 O + ] [A – ] [H 2 O] [HA] K c [BH + ] [OH – ] [H 2 O] [B] The extent to which the weak electrolytes dissociates is expressed by either ionization constants or degree of ionization. K A [ H + ] [ A – ] [ HA ] K c [H 2 O] = K B [ BH + ] [ OH – ] [ B ]](https://image.slidesharecdn.com/01solutionselectrolytesprotolyticequilibria-110914014716-phpapp02/85/01-solutions-_electrolytes__protolytic_equilibria-35-320.jpg)


![The second parameter that expresses the dissociation (ionization) of weak acids and weak bases is the degree of ionization α c (the percentage of ionization). It is found from the concentration of H + (in acid solutions, of OH – in solutions of bases) and the total concentration of the acid or base c total that is equal to the sum of both ionized and non-ionized molecules [H + ]+[HA] or [OH – ]+[BH]. For a monoprotic weak acid α c = [ H + ] c total The index specifies the total concentration, the value of α varies with the c total . The more dilute the solution, the larger is the degree α c . Because [H + ] = α c c tota l , then K A = c total α 2 1 – α If the degree of ionization is low ( α c < 0.10, i.e. 10 %), the relation is simpler: K c total α c 2 and α c ( K / c total ) ½ .](https://image.slidesharecdn.com/01solutionselectrolytesprotolyticequilibria-110914014716-phpapp02/85/01-solutions-_electrolytes__protolytic_equilibria-39-320.jpg)
![Calculation of pH values in solutions of weak acids and bases Solutions of weak monoprotic acids The total acid concentration c total = [H + ] + [HA] . Because [H + ] = [A – ] (molecules of acid gives the same number of both), [H + ] [A – ] = [H + ] 2 . Now an approximation is induced: [HA], the concentration of undissociated molecules ( c total – [H + ]), is close to c total , when the dissociation degree α c is less than 0.10 (the minute difference of the denominator can be neglected). Then , from which and in logarithmic form log [ H + ] = ½ log K A + ½ log c total The pH value of a weak acid solution HA + H 2 O H 3 O + + A – K A [ H + ] [ A – ] [ HA ] K A [ H + ] 2 c total [ H + ] = pH = ½ p K A – ½ log c total](https://image.slidesharecdn.com/01solutionselectrolytesprotolyticequilibria-110914014716-phpapp02/85/01-solutions-_electrolytes__protolytic_equilibria-40-320.jpg)
![pOH = ½ p K B – ½ log c B Solutions of weak bases The total base concentration c total = [BH + ] + [B] . Because [BH + ] = [OH – ] (molecules of base gives the same number of both), [BH + ] [OH – ] = [OH – ] 2 . The approximation as for acids: [B], the concentration of undissociated base ( c total – [OH – ]), is close to c total , when the dissociation degree α c is less than 0.10 (the minute difference of the denominator can be neglected). Then , from which and in logarithmic form log [ OH – ] = ½ log K B + ½ log c total The pH value of a weak base solution B + H 2 O BH + + OH – K B [ BH + ] [ OH – ] [ B ] K B [ OH – ] 2 c total [ OH – ] = pH = 14 – ½ p K B + ½ log c B](https://image.slidesharecdn.com/01solutionselectrolytesprotolyticequilibria-110914014716-phpapp02/85/01-solutions-_electrolytes__protolytic_equilibria-41-320.jpg)