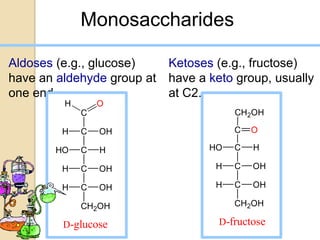
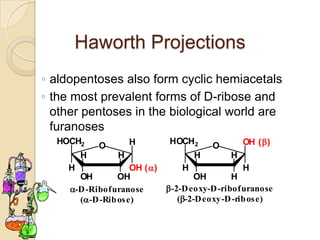
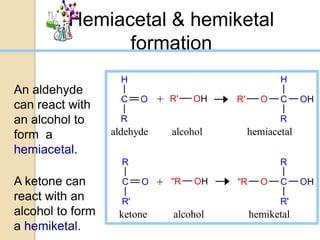
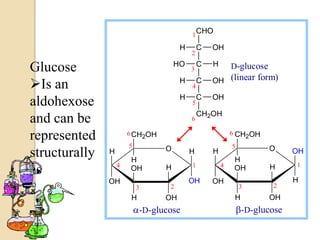
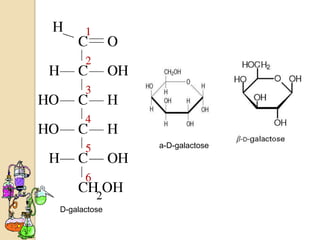
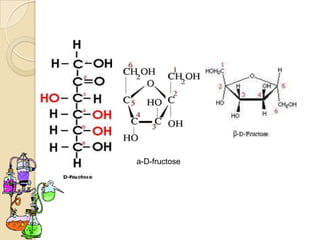
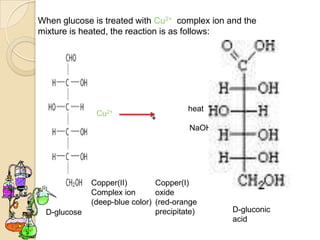
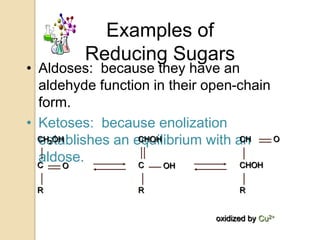
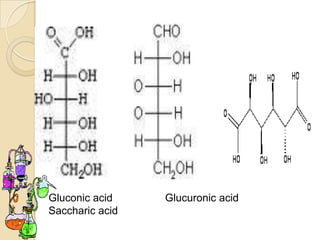
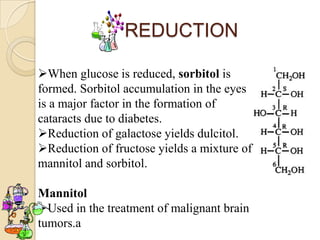
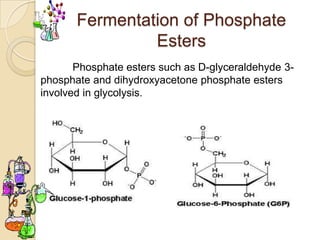
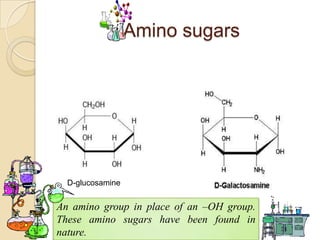
Monosaccharides are simple sugars that serve as building blocks for other carbohydrates. They include pentoses with 5 carbons like ribose and hexoses with 6 carbons like glucose and fructose. Glucose and fructose are isomers with the same formula but different structures. Monosaccharides can exist as linear molecules or in ring forms like furanoses. They undergo various reactions including oxidation, reduction, and fermentation.