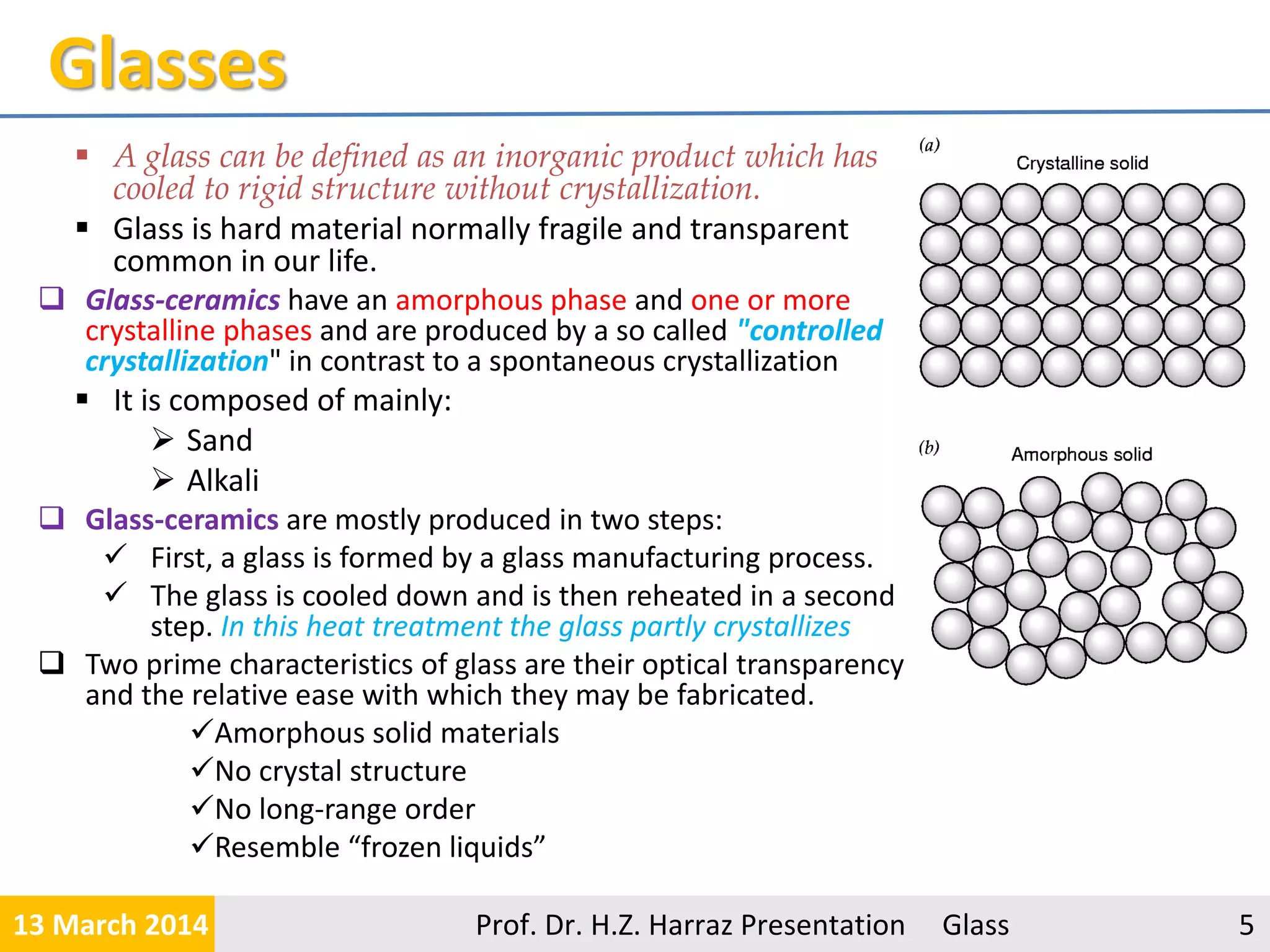
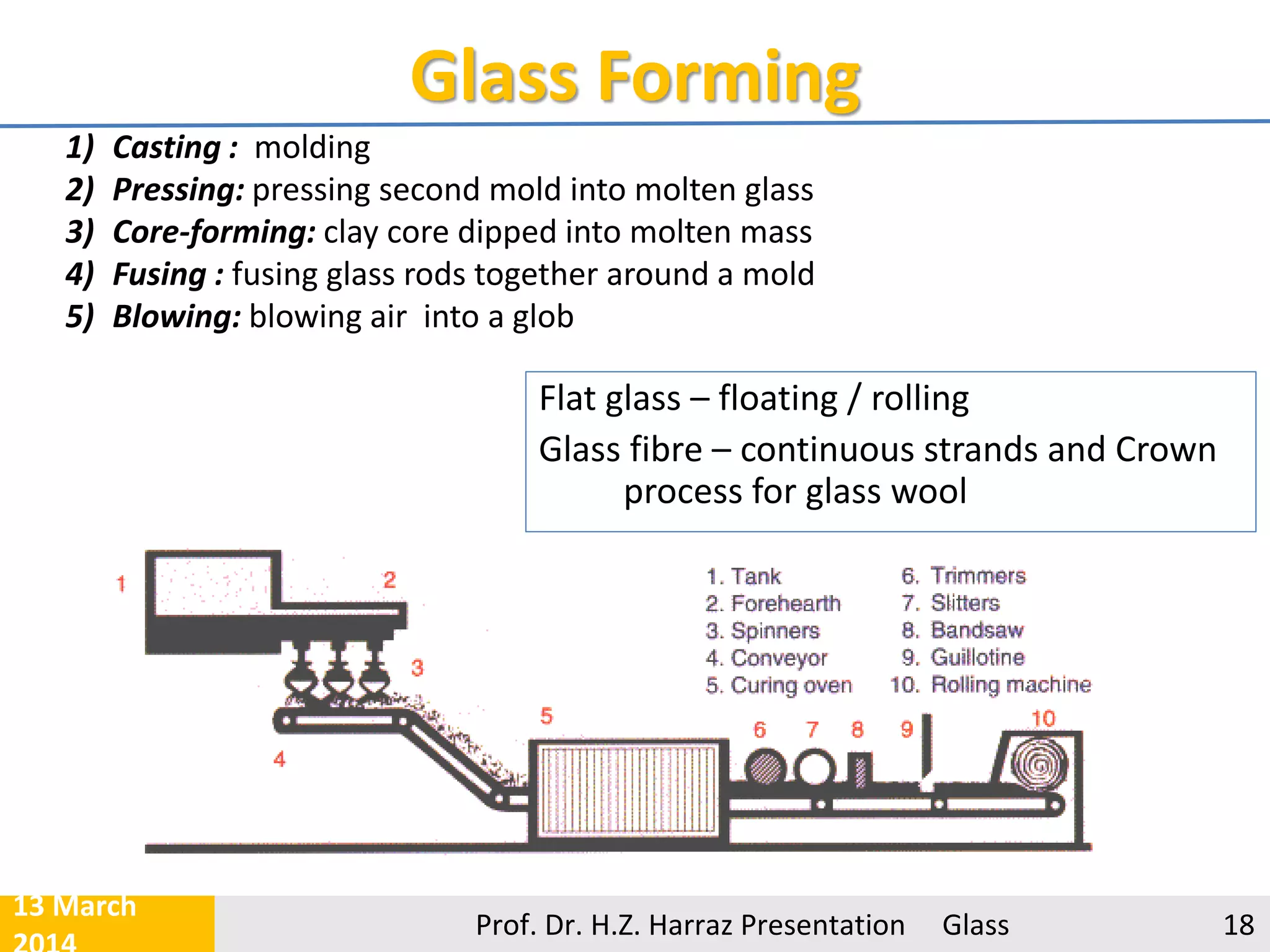
The document provides a comprehensive overview of glass, detailing its definition as an inorganic, non-crystalline material and discussing its various raw materials, manufacturing processes, and types. Key components include silica sand, limestone, and soda ash, with an emphasis on the significance of purity and specific properties in glass production. The presentation defines five major types of glass, such as soda-lime and borosilicate glasses, each with unique characteristics and applications.