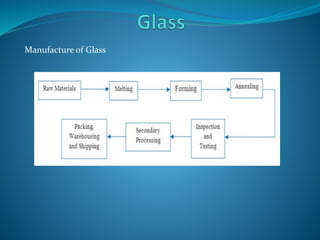
Glass is a hard, brittle substance that is typically transparent or translucent. It has no definite melting point, but softens and becomes viscous above its glass transition temperature. Chemically, glass is a non-crystalline fused mixture of silicates and other compounds. Glass is characterized by its hardness, transparency, refractive index, and low thermal and electrical conductivity. Common types of glass include soda-lime glass, borosilicate glass, and fiberglass. Glass is manufactured through heating raw materials such as silica sand, soda ash, and limestone, then rapidly cooling the melted substance. Proper annealing is required to relieve internal stresses from the manufacturing process.