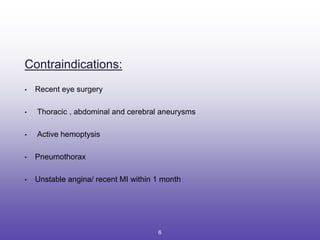
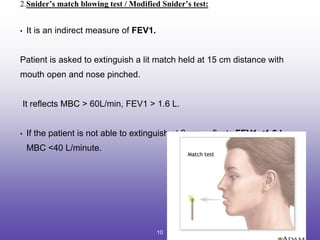
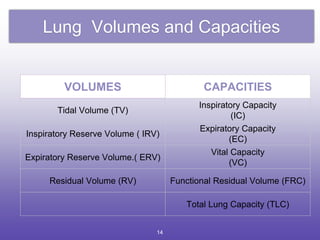
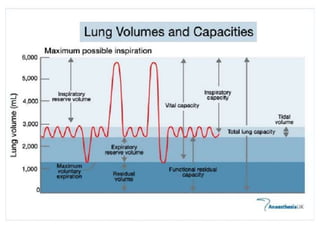
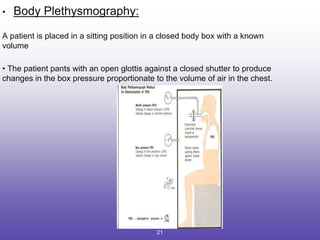
This document provides an overview of pulmonary function tests (PFTs). It discusses:
1. The classification and types of PFTs including mechanical ventilatory functions tests, lung volumes/capacities tests, spirometry, and gas exchange function tests.
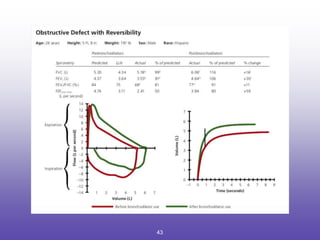
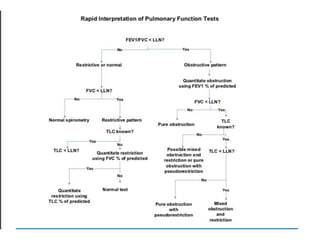
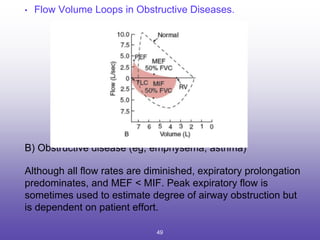
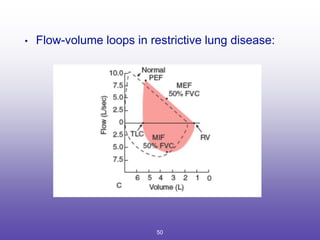
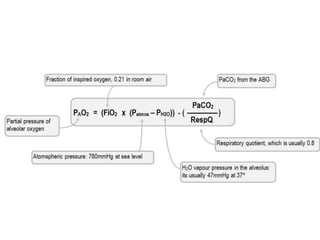
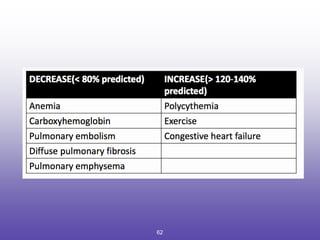
2. How to interpret PFT results including evaluating the FEV1/FVC ratio to determine obstruction and evaluating the FVC to identify restriction.
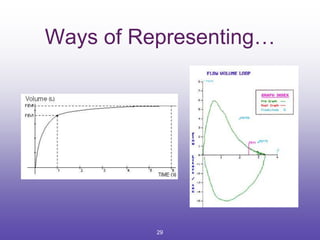
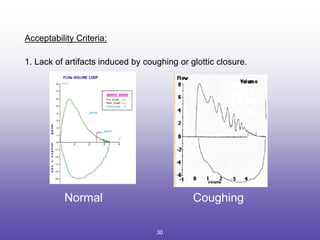
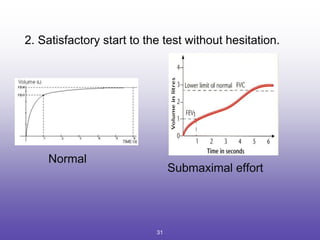
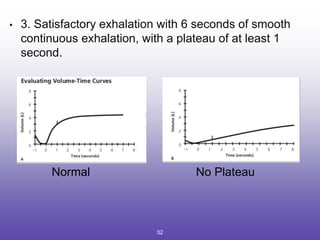
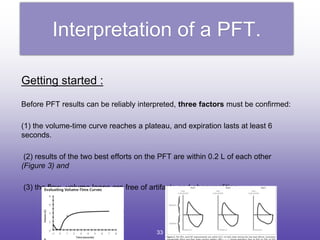
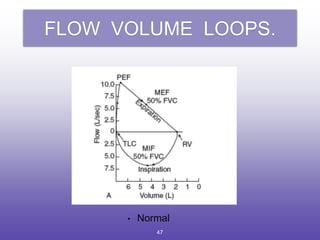
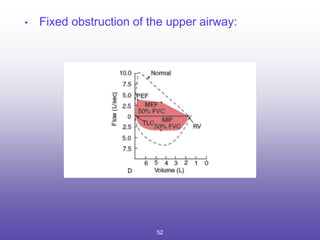
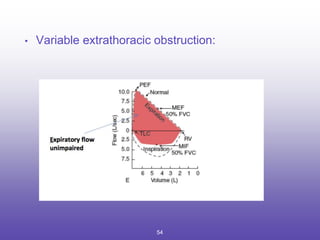
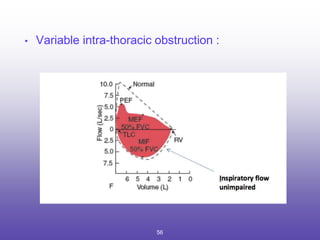
3. Key aspects of spirometry like acceptability criteria, interpreting volume-time and flow-volume curves, and confirming restrictive patterns.