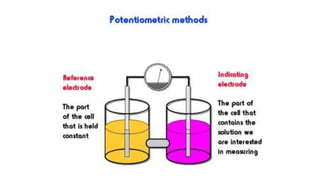
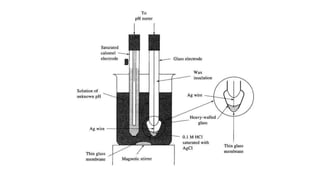
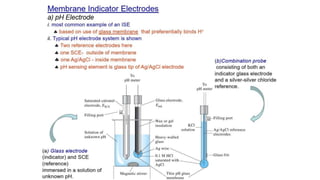
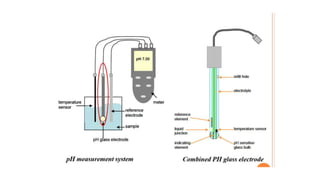
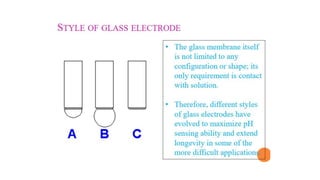
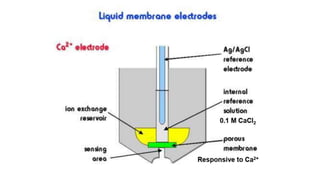
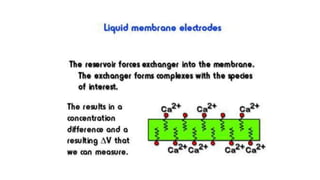
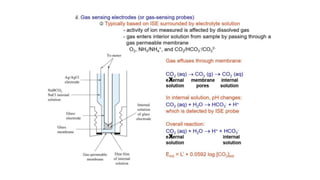
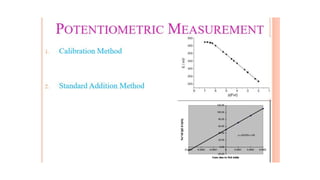
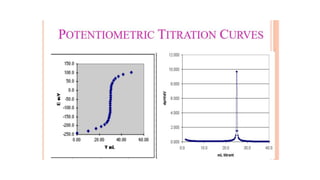
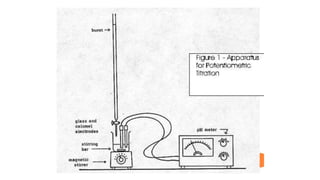
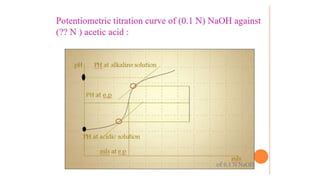
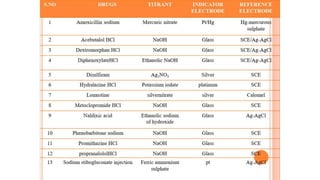
Potentiometric titration is a chemical analysis technique that relies on measuring the electromotive force (EMF) of a solution using indicator and reference electrodes. The EMF depends on the ions present in the solution. During titration, the EMF is measured after each addition of titrant and graphed versus volume added. There are four main types of potentiometric titration: acid-base titration determines concentration by neutralization, redox titration involves a redox reaction, complexometric titration forms colored complexes, and precipitation titration forms an insoluble precipitate.