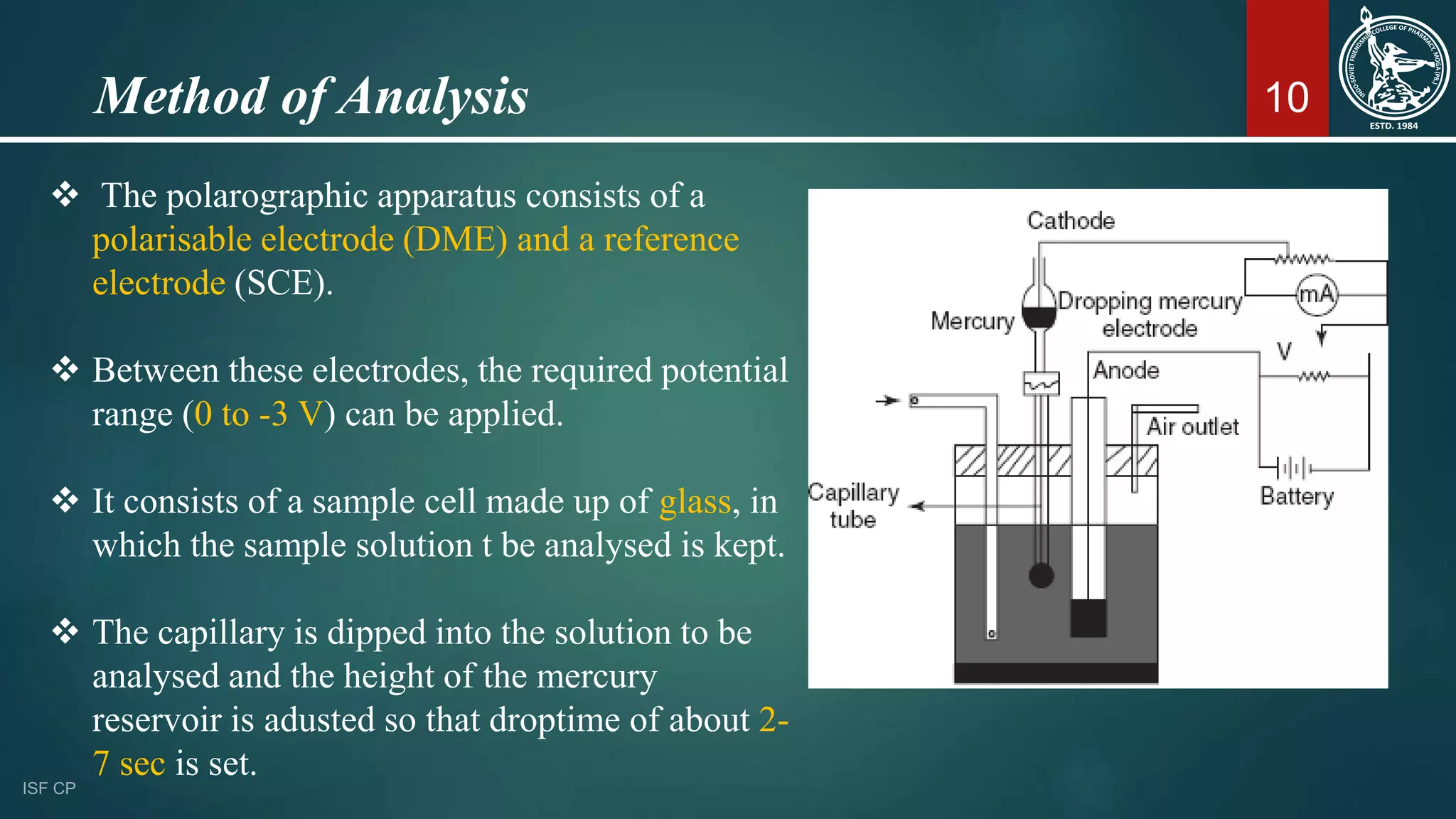
This document discusses polarography, an electromechanical technique for analyzing electroreducible or oxidizable elements using current-voltage measurements between electrodes. It covers the principles, advantages, and methodology, including the use of mercury electrodes, factors affecting diffusion current, and various applications in pharmaceuticals. The technique allows for both qualitative and quantitative analysis, enabling the detection of low concentrations of substances without prior separation.