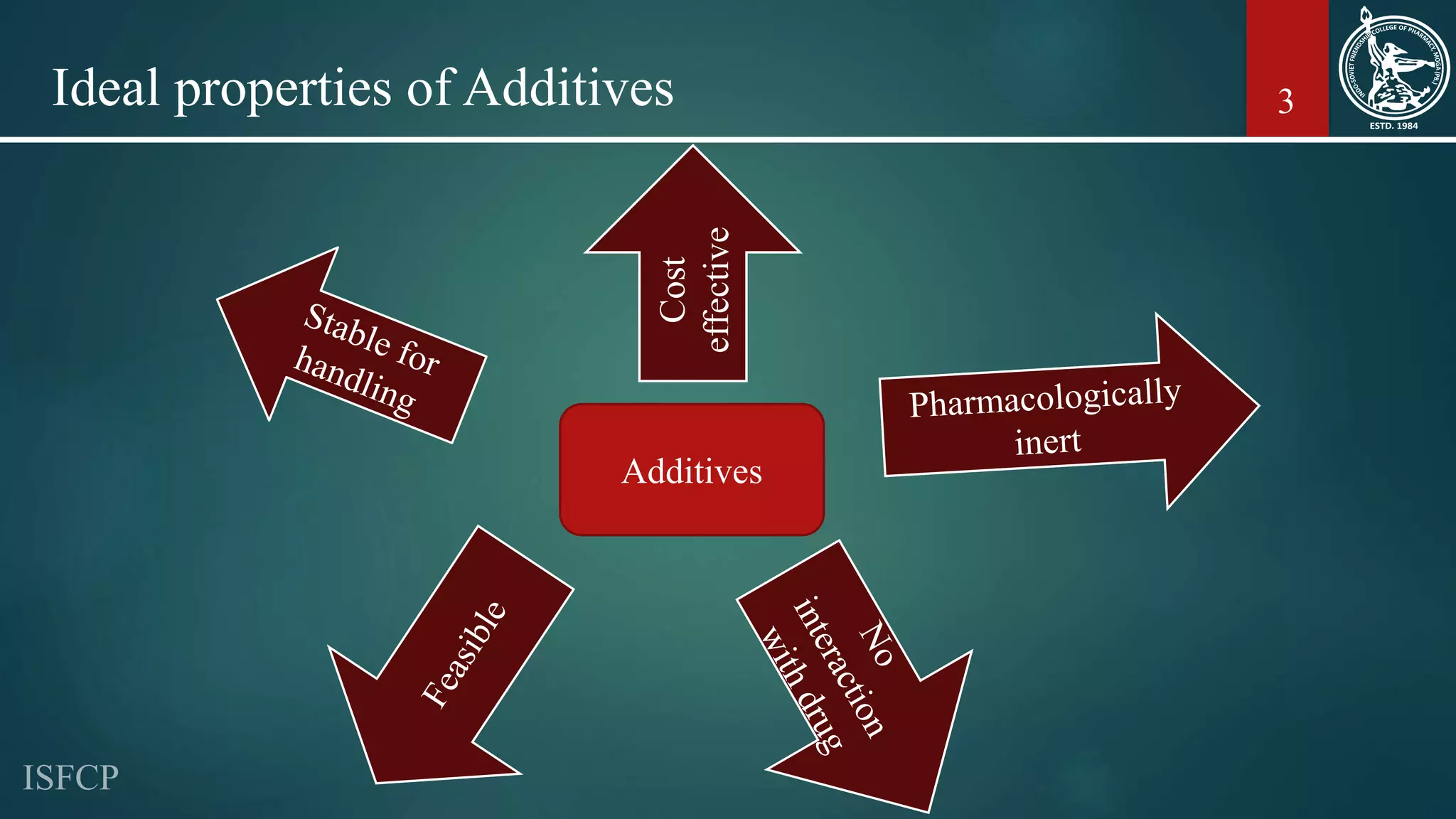
The document outlines the various types and roles of pharmaceutical additives used in drug formulations, detailing their purposes such as providing bulk, aiding absorption, and ensuring stability. It categorizes additives into fillers, binders, disintegrants, coatings, and several other functions, explaining the properties and examples of each. Additionally, the document discusses the functions of antioxidants, preservatives, sweeteners, flavoring agents, and surfactants in enhancing drug formulations.