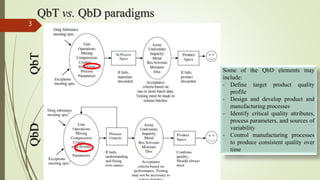
This document discusses techniques for scaling up solid dosage form production from pilot plants. It covers key steps like material handling, blending, granulation, drying, particle size reduction, slugging, compression, coating and capsule filling. For each step, parameters important for process control are identified, such as equipment type, material properties, loading amounts, time, temperature and humidity settings. The goal of scaling up is to control these parameters to consistently produce quality products at larger volumes.