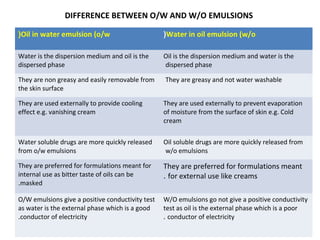
This document discusses pharmaceutical emulsions. It defines an emulsion as consisting of two immiscible liquids, one dispersed as droplets in the other. Emulsions are stabilized by emulsifying agents. Pharmaceutical applications of emulsions include oral, rectal, topical, and intramuscular administration. Different types of emulsions like O/W and W/O are described. Factors affecting emulsion stability and types of instabilities are outlined. Methods for emulsification, preservation, and packaging are also summarized.