Embed presentation
Downloaded 157 times


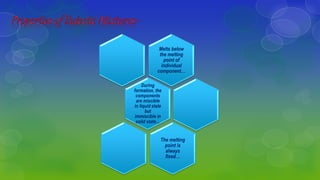
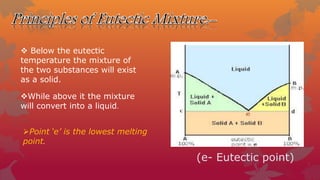






Eutectic mixtures are formed when components are miscible in liquid but immiscible in solid state, with a fixed melting point at the eutectic temperature. To create these mixtures, the ratio of components must be adjusted and impurities controlled, allowing for effective drug design and delivery. They are also used to identify compounds with similar melting points and in local anesthetics for children.








