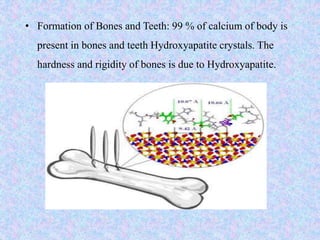
Calcium and phosphorus are important minerals that make up bones and teeth and are involved in many metabolic processes. Calcium homeostasis is regulated by parathyroid hormone, calcitonin, and vitamin D which act on the intestines, kidneys, and bones. Hypocalcemia can result from hypoparathyroidism, vitamin D deficiency, or renal disease and causes tetany and muscle spasms. Hypercalcemia has causes like hyperparathyroidism or cancer and symptoms of nausea, constipation, and renal problems. Phosphorus is found in bones and tissues and is needed for energy metabolism and cell signaling. Its absorption is regulated similarly to calcium. Hypophosphatemia can be caused by