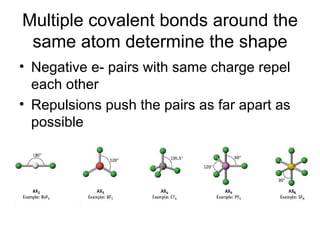
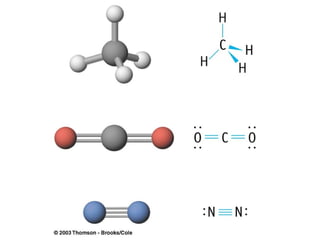
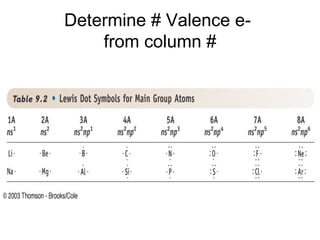
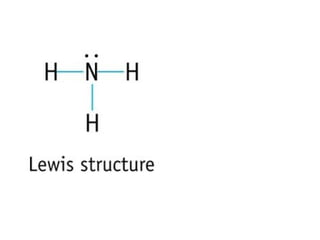
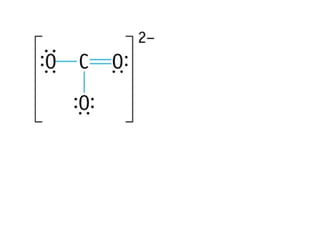
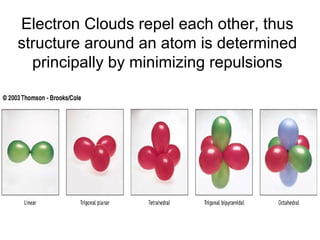
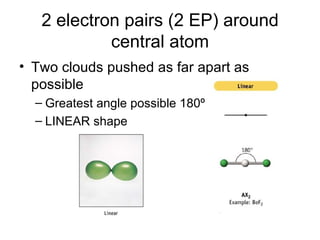
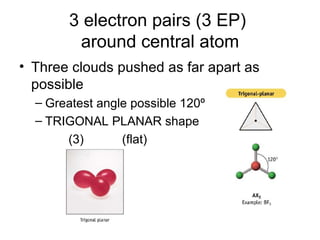
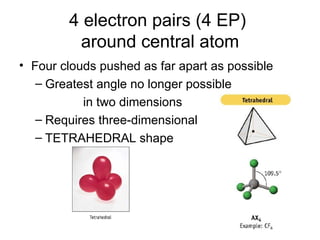
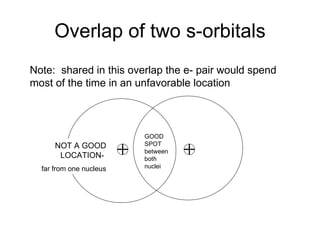
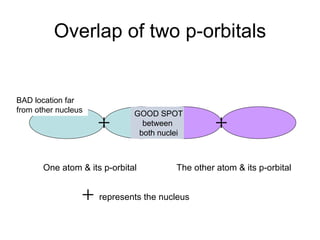
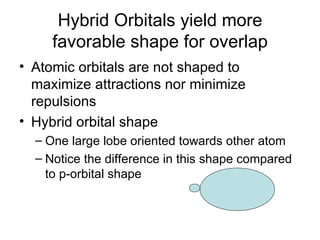
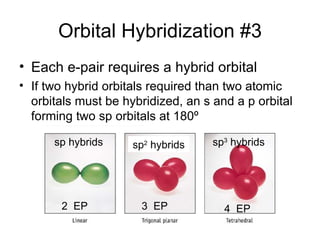
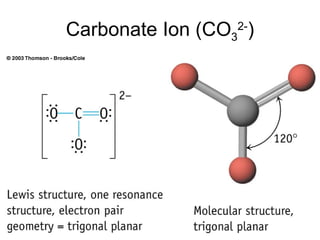
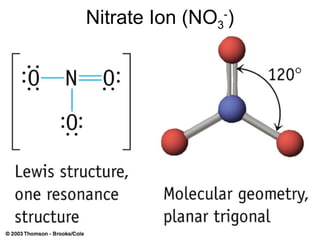
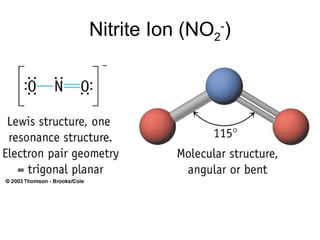
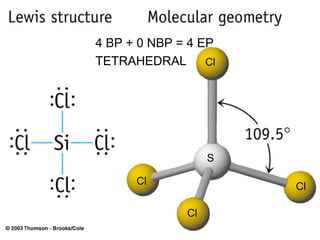
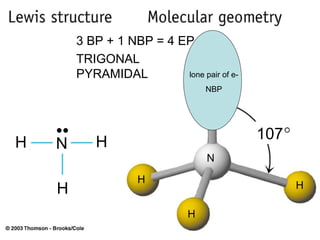
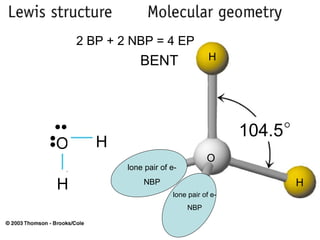
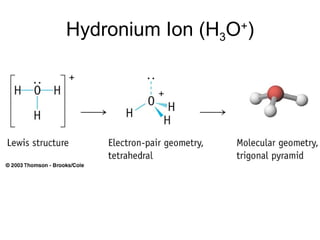
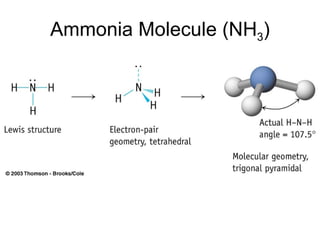
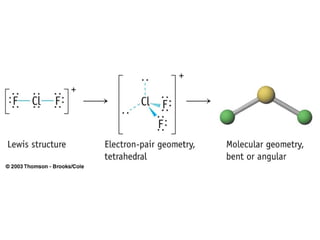
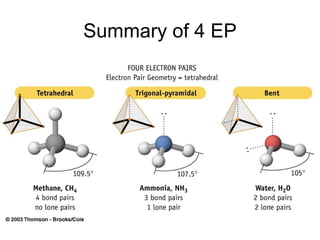
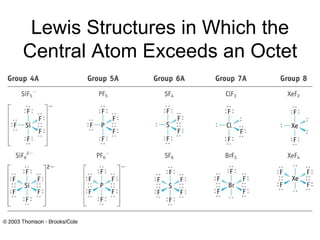
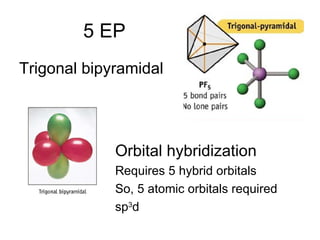
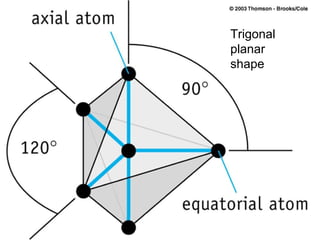
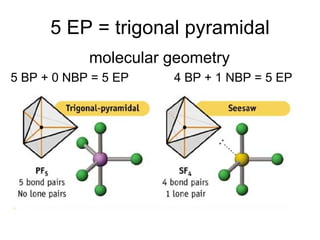
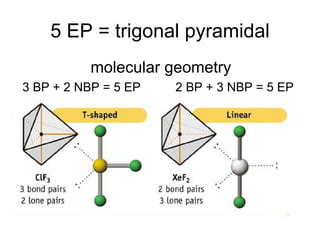
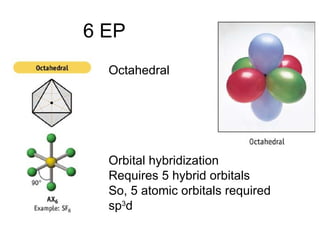
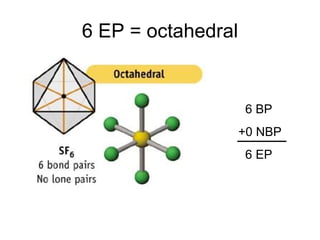
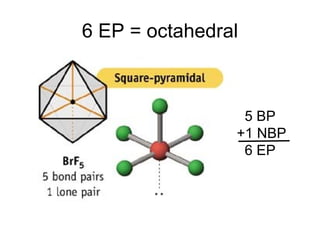
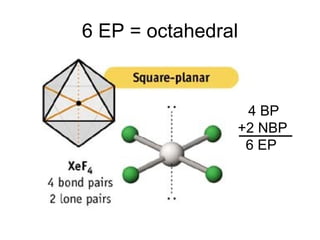
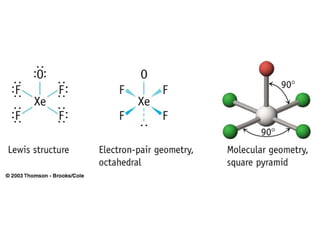
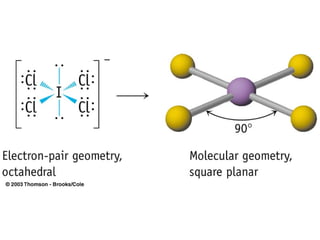
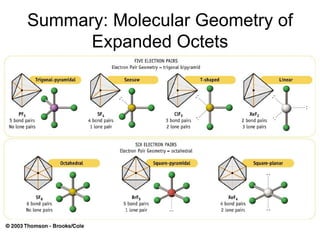
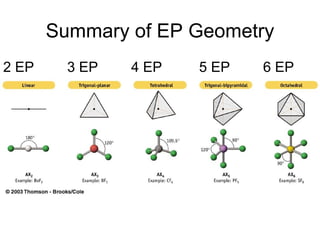
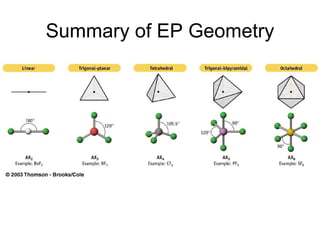
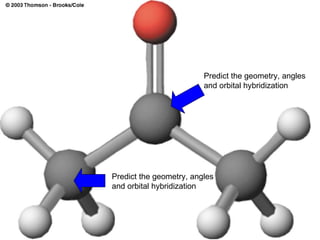
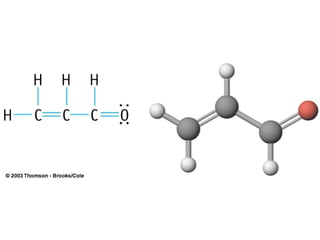
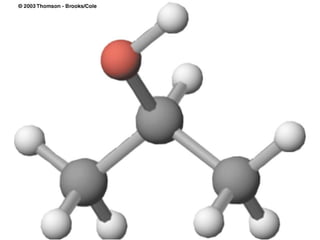
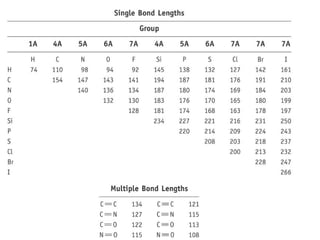
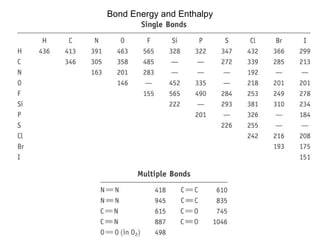
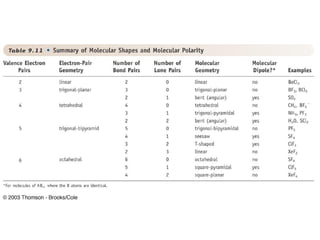
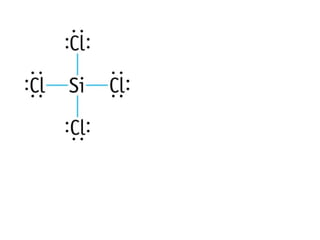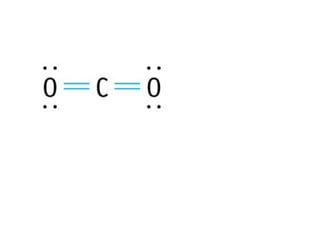The document discusses the VSEPR (Valence Shell Electron Pair Repulsion) model for understanding molecular geometry. It explains that chemical bonds form when collections of atoms are more stable and lower in energy than separate atoms. The VSEPR model uses electron pairs around central atoms to predict molecular shapes and bond angles based on minimizing electron pair repulsions. Hybrid orbital theory is also introduced to explain how atomic orbitals combine to form hybrid orbitals that are better suited for bonding arrangements.