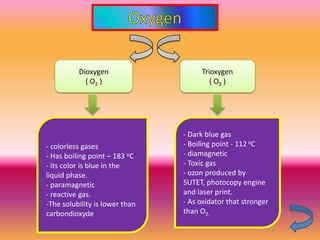
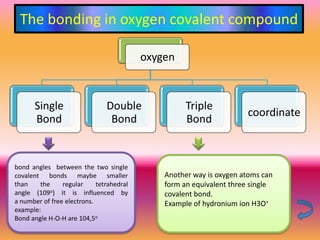
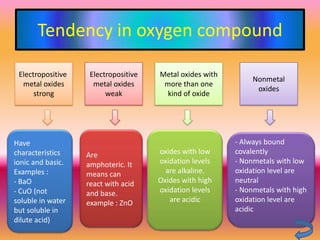
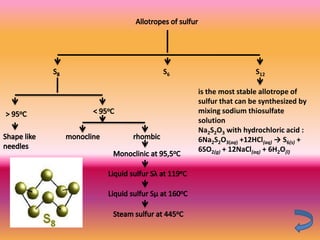
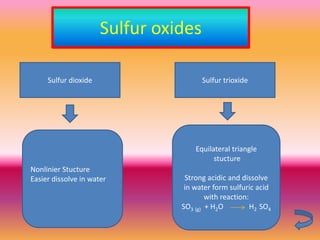
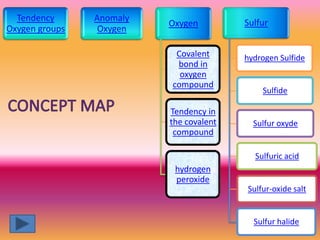
The document discusses various oxygen and sulfur compounds. It describes the characteristics of common oxygen groups like oxygen, ozone, hydrogen peroxide and their bonding structures. It also discusses the properties of sulfur and sulfur compounds such as hydrogen sulfide, sulfides, sulfur oxides, sulfuric acid, sulfur salts and sulfur halides. The document provides information on the tendencies and bonding preferences in these oxygen and sulfur compounds.
![The Preference of Oxygen Groups
Table 1. Data Of Some Oxygen Group Characteristic
elements Electron Melting Boiling point
configuration point (oC) (oC)
8O [2He] 2s2 2p4 -219 -183
16S [10Ne] 3s2 3p4 119 445
34Se [18Ar] 3d10 4s2 4p4 221 685
52Te [36Kr] 4d10 5s2 5p4 452 987
84Po [54Xe] 4f14 5d10 6s2 6p4 254 962](https://image.slidesharecdn.com/golonganoksigen-fix-111214090906-phpapp02/85/OXYGEN-3-320.jpg)