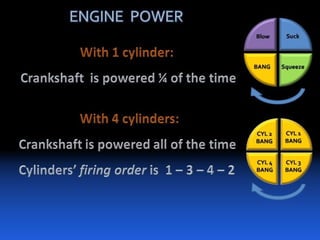
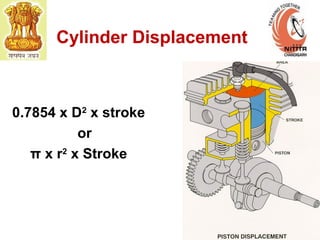
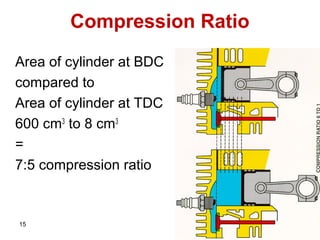
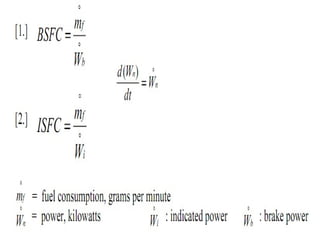
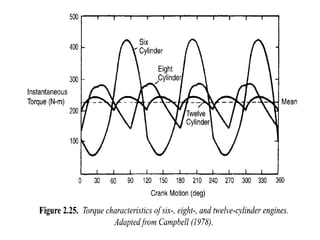
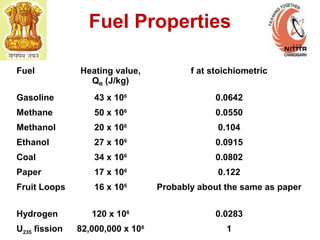
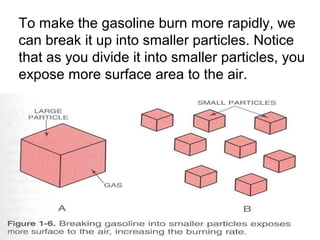
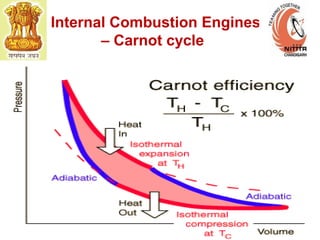
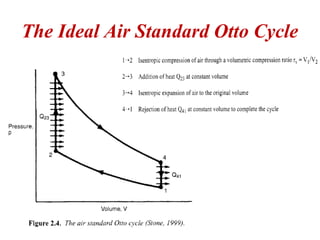
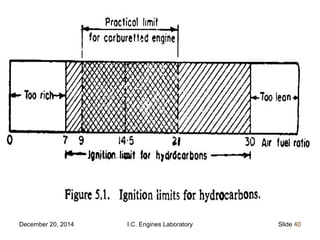
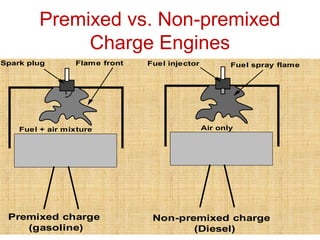
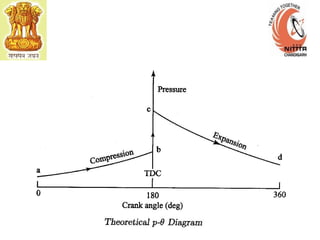
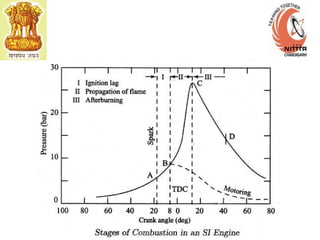
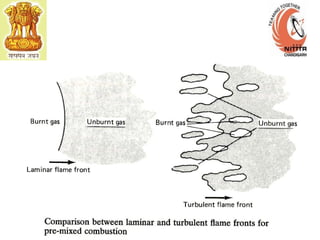
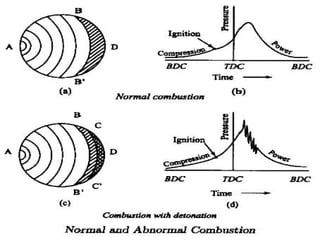
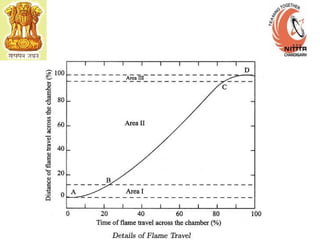
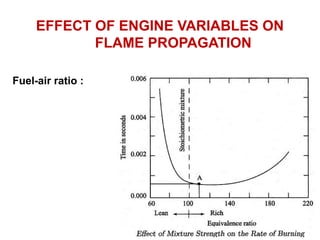
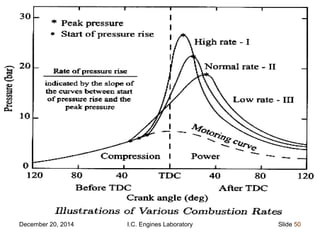
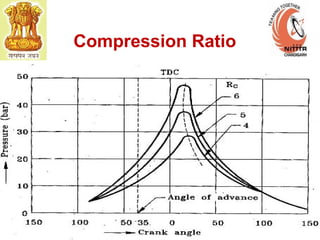
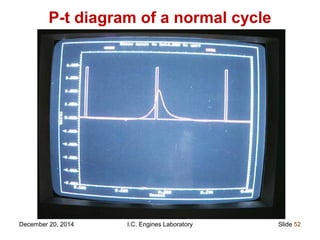
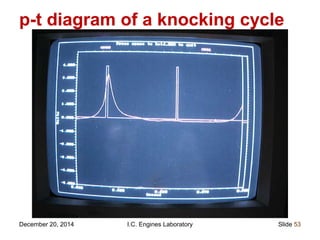
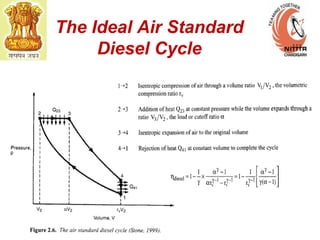
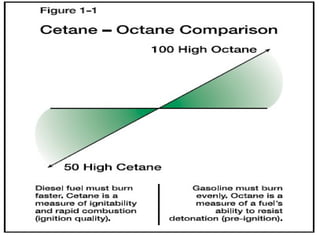
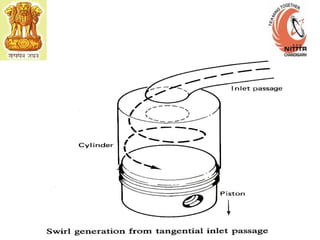
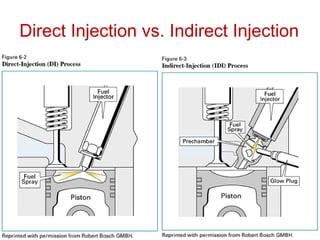
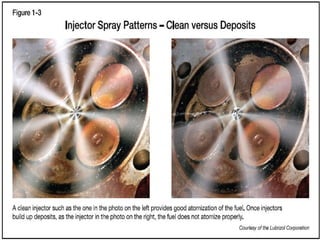
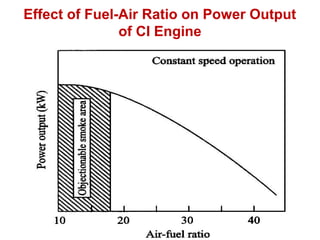
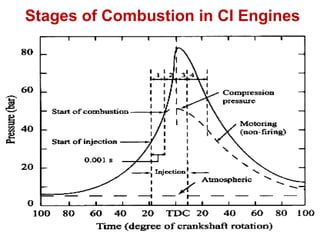
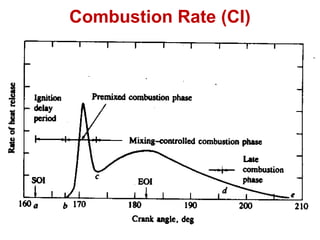
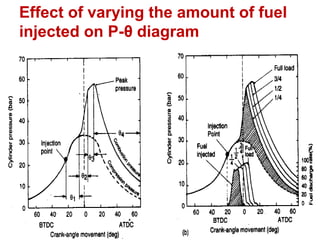
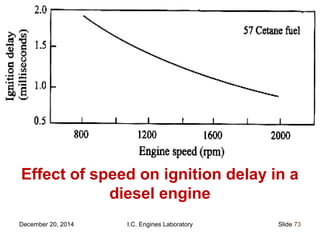
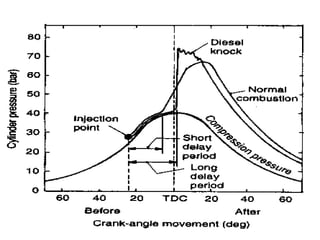
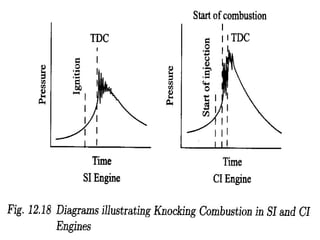
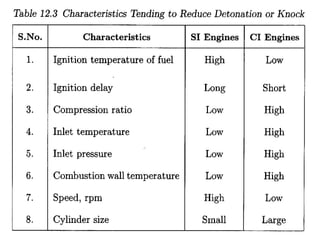
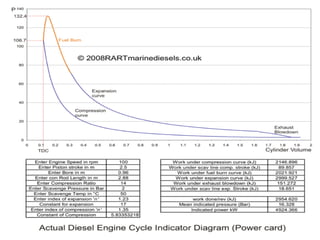
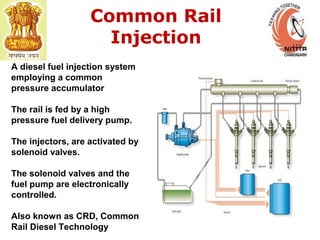
The document provides information on engine performance and terminology. It discusses how engines convert the heat of burning fuel into useful energy. It explains that engine efficiency is typically much less than 100% due to factors like friction and heat loss. It also discusses different types of engines like Otto and Diesel engines, and key engine components and metrics like bore, stroke, displacement, compression ratio, power, and torque. It provides details on fuel types, properties, and requirements for different engines. Overall, the document is a technical overview of engine performance and key concepts.